First case of hereditary xanthinuria in a Moroccan family
Aicha Ezoubeiri, Asma Labaali, Naima Fdil, Jean-François Benoist, Laila Chabaa
Corresponding author: Aicha Ezoubeiri, Laboratory of Clinical Biochemistry, Ibn Tofail Hospital, CHU MED VI, Marrakesh, 
Received: 21 Oct 2019 - Accepted: 21 Nov 2019 - Published: 13 Dec 2019
Domain: Biochemistry,Nephrology
Keywords: Hypo-uric acid, hypoxanthine, xanthine, xanthine oxidase, hereditary xanthinuria
© Aicha Ezoubeiri et al. PAMJ Clinical Medicine (ISSN: 2707-2797). This is an Open Access article distributed under the terms of the Creative Commons Attribution International 4.0 License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Cite this article: Aicha Ezoubeiri et al. First case of hereditary xanthinuria in a Moroccan family. PAMJ Clinical Medicine. 2019;1:55. [doi: 10.11604/pamj-cm.2019.1.55.20713]
Available online at: https://www.clinical-medicine.panafrican-med-journal.com//content/article/1/55/full
First case of hereditary xanthinuria in a Moroccan family
Aicha Ezoubeiri1,&, Asma Labaali1, Naima Fdil2, Jean-François Benoist3, Laila Chabaa2,4
1Laboratory of Clinical Biochemistry, Ibn Tofail Hospital, Chu Med VI, Marrakesh, Morocco, 2Department of Biochemistry, Faculty of Medicine and Pharmacy, University Cadi Ayyad, Marrakesh, Morocco, 3Department of Biochemistry and Hormonology, Robert Debré Hospital, Paris, France, 4Laboratory of Clinical Biochemistry, Arrazi Hospital, Chu Med VI, Marrakesh, Morocco
&Corresponding author
Aicha Ezoubeiri, Laboratory of Clinical Biochemistry, Ibn Tofail Hospital, CHU MED VI, Marrakesh, Morocco
Xanthinuria, which was first described in 1954 by Dent and Philpot [1], is a rare autosomal recessive disease. It is characterized by excretion of large amounts of xanthine in the urine and a tendency to form xanthine stones. Hereditary xanthinuria is a purine metabolism disorder due to inherited deficiency of the xanthine oxidoreductase (XOR) [2]. This enzyme catabolizes the oxidation of hypoxanthine to xanthine and xanthine to uric acid. Enzyme deficiency is the cause of an increase in the urinary excretion of xanthine and hypoxanthine, with a xanthine/hypoxanthine ratio of 4/1 [2,3]. Xanthine (oxydase) dehydrogenase deficiency can be isolated or associated with an aldehyde oxidase deficiency, defining respectively hereditary xanthinuria type I and II [4]. The human gene encoding for XOR is located at chromosome position 2p22, while that encoding aldehyde oxidase is at 2q33 [4-7]. Both types are characterised by plasma uric acid concentrations below 5mmol/l and plasma xanthine concentrations over 10 mmol/l. Urinary excretion of uric acid is low or undetectable, and that of xanthine is high. Less than half of the affected people have symptoms, which are caused by deposition of xanthine in the urinary tract. This results in haematuria or renal colic and rarely in acute renal failure or chronic complications related to urolithiasis. Muscle pains caused by xanthine deposition occur in a minority of cases [5]. In this work we describe the first cases of hereditary xanthinuria in a Moroccan family.
Case report: AS, a 49-year-old female from the center of Morocco, with type 2 diabetes, was directed to the laboratory of clinical biochemistry for the first time for her periodic biological monitoring. All measured biochemical parameters were normal except a moderate hyperglycemia and an undetectable serum uric acid (Table 1). The uric acid assay carried out on a second sample taken two months later gave the same result. The urinary uric acid was also undetectable. Faced with this major hypouricemia, we suspected a hereditary xanthinuria. Urine oxipurines, which were analysed by high-performance-liquid-chromatography (HPLC), revealed an increase of hypoxanthine and xanthine, together with a very low excretion of uric acid, thus confirming the diagnosis of hereditary xanthinuria (Figure 1, Table 2). Our patient was the eldest of a family of four children, born from non-consanguineous parents. Urine samples from the younger brother and sister were obtained. Urine uric acid and oxipurines were normal for the younger sister; however, the profile of the younger brother was typical of hereditary xanthinuria, with an accumulation of xanthine and normal hypoxanthine concentration (Figure 2, Table 3).
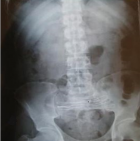
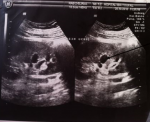
Clinically, the brother, who was 43 years old, did not show any sign of xanthinuria. The patient (AS) reported no history of kidney stones, no neurological and muscle signs and no arthritis symptoms. The clinical examination and psychomotor development were normal. The patient was then referred to the Nephrology Department. A radiographic assessment was performed in search of kidney stones including urinary tract without preparation (AUSP), renal ultrasound and pelvic CT scans. The radiography of the plain urinary tract did not objectify radiopaque urolithiasis (Figure 3). A complementary reno-bladder ultrasound showed a left calicielle dilatation without any identifiable obstacle (Figure 4). The CT urography (reno-bladder CT with contrast product injection) did not reveal any urinary calculus, but a dilated left calicielle, probably of hypotonic origin. For the younger brother (AM), the radiological analysis was not performed because he did not turn up.
Biochemical analysis: uric acid in plasma and urine were measured using a specific enzymatic method adapted for an auto-analyzer (Cobas Integra 400 plus Analyzer, Roche). Oxipurines analysis was performed by high-performance-liquid-chromatography coupled with a photodiode array detector (Waters, France) using a C18 column. The method allows the qualitative and quantitative determination of purine and pyrimidine bases. Molecules were identified by the analysis of the complete spectrum (190-320nm) and the comparison with reference spectra contained in the library and quantified at 254nm. This technique is applied to the analysis of purine and pyrimidine bases in plasma, Cerebrospinal fluid (CSF) and urine.
Here, we report the first cases of xanthinuria in a Moroccan family. The disorder was demonstrated by undetectable concentrations of uric acid in the serum and high excretion of xanthine in the urine. Both cases were clinically asymptomatic at the time of diagnosis of the pathology. Our patient presents an accumulation of xanthine and hypoxanthine with xanthine/hypoxanthine ratio superior to 3.5. While his brother presents an accumulation of xanthine and hypoxanthine, with a xanthine/hypoxanthine ratio that is more than 6.5. For our two patients, hereditary xanthinuria was confirmed, but we cannot determine whether it´s type I or II because the allopurinol loading test was not done [8]. The radiological examination of our patient was normal except for a left calicielle dilatation without any identifiable obstacle. Six months after the diagnosis of hereditary xanthinuria, she had myalgias. These muscle pains worsened progressively and forced her to abandon her work as a housekeeper. Her diabetes was unbalanced with glycated hemoglobin up to 8% which constitutes a second risk factor predisposing to the development of renal failure. So far, about 150 patients with classical xanthinuria have been described worldwide [9]. The low concentration of uric acid in the serum and high excretion of xanthine in the urine can be used as a preliminary biochemical diagnostic marker of this disease. The xanthinuria is often latent and fortuitous discovery in front of a hypouricemia or during family screening are frequent [10,11]. In 30 to 40% of cases, the disease is revealed by urinary calculus [4,12,13] that can be prevented by a diet low in purines and animal protein with sufficient water intake. The more frequent clinical manifestations of urolithiasis in children are abdominal pain (44%), hematuria (38%), fever (15%) and other symptoms secondary to urinary tract infection [14]. Arthralgia, arthritis and myalgia were also described [11,15].
These complications are explained by the low solubility of xanthine causing precipitation of xanthine crystals in the kidney, the urinary tract, joints and muscles [4,6]. Xanthine dehydrogenase (XDH) deficiency can be isolated or associated with an aldehyde oxidase deficiency, defining respectively hereditary xanthinuria type I and II [3]. A clinically distinct third type of xanthinuria due to molybdenum cofactor deficiency is characterized by the lack of sulfite oxidase activity combined with xanthine dehydrogenase and aldehyde oxidase activities [9]. The types I and II of xanthinuria can be distinguished by allopurinol loading test. Since aldehyde oxidase converts allopurinol to oxipurinol, type II but not type I xanthinuria patients lack the ability to produce oxipurinol from allopurinol [8,16]. The determination of the activity of xanthine oxidase allows confirmation of the diagnosis. It is measured by biochemical, molecular or histological analysis, usually on duodenal biopsies, rarely liver, kidney or skin biopsies [11]. Molydenum cofactor deficiency can be identified by amino acids and oxipurines analysis and is biochemically characterized by low uric acid with high oxipurines and the presence of S-sulfocysteine with low 1/2 cystine. Hereditary xanthinuria is caused by mutations in genes xanthine dehydrogenase (XDH, 2p23.1) or in one of the genes involved in molybdenum cofactor biosynthesis (Mocos, 18q12.2). Xanthine oxidase provides other physiological functions. It is involved in iron absorption in the intestine and liberation of ferritin in the liver [17,18]. Stevens et al. [19] have demonstrated antimicrobial activity of xanthine oxidase in maternal milk which contains a significant amount of this enzyme. It also has an inhibitory effect on viral growth [20].
In conclusion, we report two first cases of hereditary xanthinuria in a Moroccan family. In our patient the risk of developing renal failure is great, because of her unbalanced diabetes and the hereditary xanthinuria. The number of cases of hereditary xanthinuria declared worldwide is probably underestimated because of the fact that this pathology is often asymptomatic.
The authors declare no competing interests.
Aicha Ezoubeiri carried out the biochemical analyses and drafting of the manuscript. Asma Labaali conducted the survey with the family of the patient and the various radiological tests. Naima Fdil helped to write the manuscript and reviewed the literature. Jean-François Benoist performed the xanthine and hypoxanthine assays and participated in drafting of the manuscipt. Laila Chabaa conceived of the study, and participated in its design and coordination and helped to draft the manuscript.
Table
1: results of standard laboratory tests of the patient
Table 2:
results of determination of urinary oxipurines of patient
Table 3:
results of determination of urinary oxipurines of patient
Figure 1: corresponds
to the chromatogram of the HPLC assay of urinary oxipurines of the patient.
The peaks of hypoxanthine and xanthine are indicated by arrows respectively at
14.895 and 16.143 minutes
Figure 2: present the
chromatogram of the HPLC assay of urinary oxipurines of the patient. The
peaks of hypoxanthine and xanthine are indicated by arrows respectively at 14.483
and 15.821 minutes
Figure 3: corresponds
to a radiological examination of the patient, radiography of the urinary
tract without preparation, in order to search the presence of kidney stones,
showing no radiopaque urinary stones
Figure 4: corresponds
to a reno-bladder echography of patient, in order to search the presence of
kidney stones, showing a left calicielle dilatation indicated by arrows
- Dent CE, Philpot GR. Xanthinuria: an inborn error of metabolism. Lancet. 1954;266(6804):182-185. PubMed | Google Scholar
- Beaudet AL, Sly WS, Valle D. The metabolic and molecular basis of inherited disease. Reviewed in Simmonds HA, Reiter S, Nishino T. Hereditary xanthinuria. Chapter 54 7th ed New York: Mc Graw-Hill. 1995;1781-97.
- Gok F, Ichida K, Topaloglu R. Mutational analysis of the xanthine dehydrogenase gene in a Turkish family with autosomal recessive classical xanthinuria. Nephrol Dial Transplant. 2003;18(11):2278-83. PubMed | Google Scholar
- Mayaudon H, Bauduceau B, Dudy O, Cepa F, Roul G, Burnat P. Acide urique indosable dans le sang et les urines: déficit en xanthine déshydrogénase (ou xanthiurie héréditaire). Rev Med Int. 1999;20:445-9. Google Scholar
- Raivio KO, Saksela M, Lapatto R. Xanthine oxidoreductase- Role in human pathophysiology and hereditary xanthinuria. The metabolic and molecular basis of inherited disease. 8e ed New York: McGraw Hill. 2001;2653-62. Google Scholar
- Bordier L, Blanchard A, Sarret D, Herody M, Nedelec G, Duvic C. Hypo-uricémie, un vieux sujet et de nouveaux concepts. Presse Med. 2004 Apr 26;33(8):555-63. PubMed | Google Scholar
- Reiter S, Simmonds HA, Zoller N, Braun S, Knedel M. Demonstration of a combined deficiency of xanthine oxidase and aldehyde oxidase in xanthinuric patients not forming oxipurinol. Clin Chim Acta. 1990 Mar 15;187(3):221-34. PubMed | Google Scholar
- Ichida K, Yoshida M, Sakuma R, Hosoya T. Two siblings with classical xanthinuria type 1: significance of allopurinol loading test. Intern Med. 1998 Jan;37(1):77-82. PubMed | Google Scholar
- Stiburkova B, Krijt J, Vyletal P, Bartl J, Gerhatova E, Korinek M et al. Novel mutations in xanthine dehydrogenase/oxidase cause severe hypouricemia: biochemical and molecular genetic analysis in two Czech families with xanthinuria type I. Clinica Chimica Acta. 2012 Jan 18;413(1-2):93-99. PubMed | Google Scholar
- Mayaudon H, Burnat P, Eulry F, Payen C, Dupuy O, Ducorps M et al. La xanthinurie hereditaire, cause rare d'hypo-uricemie: 2 observations. Presse Med. 1998;27:661-663.
- Kawachi M, Kono N, Mineo I, Yamad Y,Tarui S. Decreased xanthine oxidase activities and increased urinary oxypurines in heterozygotes for hereditary xanthinuria. Clin Chim Acta. 1990 Apr 30;188(2):137-46. PubMed | Google Scholar
- Badertscher E, Robson WL, Leung AKC, Trevenen CL. Xanthine calculi presenting at 1 month of age. Eur J Pediatr. 1993 Mar;152(3):252-4. PubMed | Google Scholar
- Roca B, Calabuig C, Sastre J, Arenas M. Hereditary xanthinuria and Ehlers-Danlos syndrome. J Inherit Metab Dis. 1992;15(6):881-882. PubMed | Google Scholar
- Matos P, Maherzi M, Pinto E, Guighard JP. Approche pratique de la lithiase urinaire chez l'enfant. Rev Med Suisse Romande. 1994 Oct;114(10):889-94. PubMed
- Bory C, Chantin C, Boulieu R. Comparison of capillary electrophoretic and liquid chromatographic determination of hypoxanthine and xanthine for the diagnosis of xanthinuria. J Chromatograph A. 1996 Apr 12;730(1-2):329-31. PubMed | Google Scholar
- Yamamoto T, Higashino K, Kono N, Kawachi M, Nanahoshi M, Takahashi S et al. Metabolism of pyrazinamide and allopurinol in hereditary xanthine oxidase deficiency. Clin Chim Acta. 1989 Feb 28;180(2):169-75. PubMed | Google Scholar
- Topham RW, Walker MC, Calish MP. Liver xanthine dehydrogenase and iron mobilization. Biochem Biophys Res Commun. 1982 Dec 31;109(4):1240-1246. PubMed | Google Scholar
- Bray RC. Molybdenum iron-sulfer flavin hydroxylases and related enzymes. In "oyer PD (Eds.), the enzymes" 3th ed, New York: Academic Press; 1975:299-419.
- Stevens CR, Miller TM, Clinch JG, Kanczler JM, Bodamyali T, Blake DR. Antibacterial properties of xanthine oxidase in human milk. Lancet. 2000 Sep 2;356(9232):829-830. PubMed | Google Scholar
- Taibi G, Paganini A, Gueli MC, Ampola F, Nicotra CM. Xanthine oxidase catalyzes the synthesis of retinoic acid. J Enz Inh. 2001;16(3):275-285. PubMed | Google Scholar