A 3-year experience with cardiac device therapy in a private cardiovascular hospital in Northern Nigeria: a retrospective descriptive study
Okorie Chinedum Anya, Andrew Kamsoko Ndakotsu, Rupert Ndubuisi Chima, Eniola Ruth Ajayi, Onomhen Leticia Imafidon, Nnamdi Lionel Nwoke, Adeyemo Jerry Adesola, Henry Rotimi Solanke
Corresponding author: Okorie Chinedum Anya, Cardiocare Multi-Specialty Hospital, Garki Area 11, Abuja, Nigeria 
Received: 06 Aug 2023 - Accepted: 27 Sep 2023 - Published: 09 Oct 2023
Domain: Cardiology,Internal medicine
Keywords: Atrioventricular block, cardiac resynchronization therapy, implantable cardioverter-defibrillator, heart failure, ventricular arrhythmias
©Okorie Chinedum Anya et al. PAMJ Clinical Medicine (ISSN: 2707-2797). This is an Open Access article distributed under the terms of the Creative Commons Attribution International 4.0 License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Cite this article: Okorie Chinedum Anya et al. A 3-year experience with cardiac device therapy in a private cardiovascular hospital in Northern Nigeria: a retrospective descriptive study. PAMJ Clinical Medicine. 2023;13:11. [doi: 10.11604/pamj-cm.2023.13.11.41344]
Available online at: https://www.clinical-medicine.panafrican-med-journal.com//content/article/13/11/full
Research 
A 3-year experience with cardiac device therapy in a private cardiovascular hospital in Northern Nigeria: a retrospective descriptive study
A 3-year experience with cardiac device therapy in a private cardiovascular hospital in Northern Nigeria: a retrospective descriptive study
Okorie Chinedum Anya1,&, ![]() Andrew Kamsoko Ndakotsu1, Rupert Ndubuisi Chima1, Eniola Ruth Ajayi2,
Andrew Kamsoko Ndakotsu1, Rupert Ndubuisi Chima1, Eniola Ruth Ajayi2, ![]() Onomhen Leticia Imafidon1,
Onomhen Leticia Imafidon1, ![]() Nnamdi Lionel Nwoke1, Adeyemo Jerry Adesola3, Henry Rotimi Solanke4
Nnamdi Lionel Nwoke1, Adeyemo Jerry Adesola3, Henry Rotimi Solanke4
&Corresponding author
Introduction: cardiac device therapy uses electronic devices implanted into the heart to improve cardiac function by correcting abnormal cardiac rhythms and coordinating cardiac contractility. They are usually indicated in heart failure and in the presence of life-threatening abnormal heart rhythms. This study aimed to report a 3-year experience with cardiac device therapy in a cardiovascular center in northern Nigeria, highlighting the most common indications for cardiac device therapy procedures, the effect of the cost of the procedure, and the post-procedure follow-up assessment.
Methods: this was a retrospective descriptive study of 48 adult patients who underwent cardiac device therapy in our cardiovascular center from January 1st, 2020, to December 31st, 2022. Data were retrieved from their electronic medical records and cardiac catheterization laboratory and subsequently analyzed.
Results: the most common procedure was dual chamber permanent pacemaker implantation, 41.6%, and the most common indication for it was a 3rd degree AV block, 50%. Heart failure with reduced EF (HFrEF) and left ventricular ejection fraction (LVEF) < 30% was the commonest indication for implantable cardioverter defibrillator insertion. Only 9 (18.7%) of the patients had the cost of their procedures covered by their health insurance plan, and only 13 (27%) could attend their 3-month post-procedure follow-up clinic visit for clinical evaluation and device interrogation.
Conclusion: the use and practice of remote cardiac device monitoring will not only improve the quality of the post-procedure follow-up assessment but can also provide a means for cutting the cost of the cardiac device implantation procedure.
Device therapy refers to the use of implantable electronic devices to improve cardiac rhythm and function [1]. The most common devices used include permanent pacemakers, implantable cardioverter defibrillators, and cardiac resynchronization therapy (CRT). One of the first cardiac devices to be used in humans was the permanent pacemaker, which came into use in the 1960s and was used to prevent bradycardia in cardiology patients who had sinus node disease or atrioventricular blocks [1]. The pacemaker device is composed of a pulse generator, which generates the electrical current needed for stimulation of the heart muscle, and the leads, or electrodes, which transmit the generated current from the pulse generator to the heart [2]. The first pacemaker was inserted in 1958 into a Swedish man who had severe Stoke-Adams syndrome and was constantly being resuscitated almost on a daily basis [3]. However, its indications have evolved to include other heart conditions that cause bradycardia or syncope, like heart failure, long QT syndrome, hypertrophic cardiomyopathy, and post-myocardial infarction bradycardia [2]. The era of clinical use of implantable cardioverter defibrillator (ICD) therapy began in the 1980s [4]. It was used for the treatment of ventricular arrhythmias, where it consistently demonstrated superiority over medical therapy in the primary and secondary prevention of sudden cardiac death [5]. These ventricular arrhythmias are poorly organized and lead to loss of cardiac muscle contraction, inadequate blood supply, and then loss of consciousness. Sudden death occurs if they persist for more than 5 minutes [6]. One component that distinguishes the ICD from the pacemaker is the presence of a 'shocking coil' [5]. This coil helps the ICD perform its function of aborting lethal rhythm by shocking the heart muscle and causing electrical cardioversion or defibrillation [7]. There is also a pacemaker function that sends electrical impulses to increase the heart rate when a slow heart rate is sensed or to counteract a tachycardia when it occurs [7].
Cardiac resynchronization therapy (CRT) is one of the most exciting recent advancements in the treatment of heart failure [8]. Whereas the pacemakers have two leads attached to the heart in the right atrium (RA) and right ventricle (RV), the CRT has a third lead, which is placed on the lateral or posterolateral wall of the left ventricle by passing it through the coronary sinus [8]. In diseased hearts, the conduction system of the ventricles can be impaired, leading to one part of a ventricle being activated or stimulated earlier or later than another part [8]. This electrical dyssynchrony leads to mechanical dyssynchrony, which can ultimately lead to muscle remodeling [8]. And with this remodeling, the right and left ventricles don't contract simultaneously, leading to decreased cardiac pumping efficiency, stroke volume, ejection fraction, and, in some cases, mitral regurgitation [8]. With the CRT, the right and left ventricles can be paced simultaneously, which increases cardiac pumping efficiency and left ventricular ejection fraction [8]. One of the earliest reports that demonstrated the efficacy of CRT was a case series done in 199 by Cazeau et al. [9]. In this study, eight subjects with end-stage heart failure and biventricular dyssynchrony had temporal biventricular pacing done for them. Results showed that the four subjects that survived 3 months after the procedure had an improvement in their New York Heart Association (NYHA) functional class, from Class IV to Class II [9]. These and other favorable early experiences set the stage for further technological development and larger trials of CRT [10].
As with many high-income countries during the past century, low-and middle-income countries, including Nigeria, are now experiencing an accelerating increase in the prevalence of cardiovascular disease. In the previous 50 years, most of the presentations of cardiovascular conditions were as a result of malnutrition and communicable diseases, with rheumatic heart disease and cardiomyopathies being the major presenting cardiovascular diseases; however, by 1999, continued improvements in economic circumstances, combined with urbanization and radical changes in the nature of work-related activities, led to dramatic changes in diet, activity levels, and behaviors such as smoking [11]. Over the next decade, more cases of heart failure from different primary conditions (mostly hypertension and cardiomyopathy) were reported and characterized, as well as an increased prevalence of cardiac arrhythmias [11]. This points to the fact that the need for cardiac device therapy in Nigeria is on the rise, and more centers that offer cardiac device implantation will need to be established. In 2018, the Pan-African Society of Cardiology (PASCAR) cardiac arrhythmia and pacing task force published a report of statistics on the use of cardiac electronic devices and interventional electrophysiological procedures in Africa from 2011 to 2016 [12]. In this report, data collected revealed that the number of functioning cardiac device implantation centers in Nigeria was 12, but that in 2013, Nigeria had the lowest rate for ICD and CRT implantation at 0.02 per million population, and in 2014, Nigeria also had the lowest permanent pacemaker (PPM) implantation rate at 0.14 implants per million population [12]. From the time this report was published until today, there has been a gradual increase in the number of centers that offer cardiac device therapy in Nigeria, and these procedures are becoming increasingly popular. This study describes a 3-year experience with cardiac device therapy in our centre in Nigeria, with the aim of highlighting the commonest challenges faced and suggesting possible solutions.
This was a retrospective descriptive study done on 48 patients who underwent cardiac device therapy at Cardiocare Hospital from the 1st of January 2020 to the 31st of December 2022.
Data collection: the data for this study was extracted from the electronic medical record (EMR) of these patients who underwent dual chamber permanent pacemaker (DCPPM), ICD, CRT-Pacemaker, and CRT-Defibrillator procedures in our center. It included patient age, sex, indication for the procedure, and present co-morbidities. The method of payment for the procedure, post-procedure complications, and follow-up data were also collected. The consent to extract these data from the electronic medical records (EMR) of these patients for this study was obtained from the Chairman of the Research Committee of the Limi Hospital Group.
Exclusion criteria: we excluded patients who had incomplete medical and procedural data and those who had their procedures completed in two or more stages.
Summary of cardiac device therapy procedures: in dual chamber permanent pacemaker insertion, central venous access is through the left subclavian vein using Seldinger's technique. Once the guidewires have reached the right atrium (RA), just above the inferior vena cava, a subcutaneous pocket is created according to the size of the device, and the patient's anatomy. Leads are then implanted by channeling them through a 'peel-away introducer sheath', which is used to replace the guidewires. Most RA leads are positioned in the lower lateral border of the RA, while right ventricular (RV) leads are positioned in the interventricular septum or RV apex. Leads are anchored by active fixation, and their position is confirmed with fluoroscopy in a left anterior oblique (LAO) projection. The other ends of the leads are then sutured to the pectoralis major muscles in the subcutaneous pockets. The process is similar for ICD implantation except the RV lead has a "shocking coil", and the pulse generator is bigger. In cardiac resynchronization therapy (CRT), whether a CRT-pacemaker (CRT-P) that consists of a RA pacemaker and biventricular pacemakers or a CRT-defibrillator that consists of the same in addition to a "shocking coil" in the RV. The left ventricular (LV) lead is placed on the outer wall of the LV by passing it through a branch of the coronary sinus (CS). Coronary sinus cannulation is usually difficult; we commonly use the conventional CRT implantation techniques (telescoping catheter system) described by Vogt J et al. [13] to cannulate the CS. The lead position is also confirmed by fluoroscopy, and the other end of the lead is sutured to the pectoralis major muscle in the subcutaneous pocket.
Lead parameters check: when a lead is positioned in a good anatomical position, we ensure the lead parameters are also satisfactory by assessing the pacing threshold, lead sensing, and slew rate, as well as the impedance, using a pacing system analyzer (PSA). A testing cable, also known as PSA cable, is connected at one end to the PSA, while at the other end, a pair of crocodile clips are used to test the lead. Once connected to the lead and before testing the parameters of the lead, the 'injury current' is checked. If it is large and positive, it suggests good contact between the lead tip electrode and the myocardium. Maximal output pacing is also performed on each lead to ensure there is no stimulation of extracardiac structures like the phrenic nerve, which is normally indicated by diaphragmatic twitching. Once the leads have been anchored, the pacemaker, ICD, or CRT generator is connected to the leads and placed in the pocket. We use two layers of absorbable sutures to close the deeper subcutaneous layer and a single layer of absorbable sutures in the subcuticular plane. Finally, after the wound has been closed with the generator and the leads in the pocket, an antiseptic dressing is placed over the wound area. Prior to moving the patient from the Cath lab table, fluoroscopy or cine recording is made to have a final view of the implanted system as well as to confirm that the leads are still stable.
Data analysis: data were analyzed with SPSS version 20.0 (SPSS, an IBM company) and reported as numbers, percentages, or means.
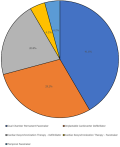
The sex distribution was 39.5% female and 60.5% male. The age range was between 36 and 86 years, with an average age of 66.1 years. Most of the patients were in their 70s (33%). The most common procedure done was the implantation of a DCPPM in 20 (41.6%) out of 48 patients, followed by ICD implantation in 14 (29.2%), and then CRT-D in 10 (20.8%). Other procedures had a frequency of less than 5% (Figure 1). Out of the 48 patients, 39 (81.2%) of them made 'out-of-pocket' payments for the procedure, including those with a health insurance plan, and only 9 (18.7%) of them had payment for the procedure covered by their health insurance. The several indications for performing each procedure are presented in Table 1. Type II Diabetes mellitus was the most common co-morbidity and was found in 11 (22.9%) of them, while other co-morbidities recorded included hypothyroidism, HIV infection, chronic kidney disease, benign prostatic hyperplasia (in the males), and prostate and breast cancer. The complication rate was 6.25% (3 out of 48), which included one lead dislodgement that occurred after a DCPPM and two implantation site infections that occurred after a DCPPM and a CRT-D implantation. There was a case of severe hypoglycemia and hypotension that complicated the implantation of an ICD in an 81-year-old with dilated cardiomyopathy and heart failure and background type II diabetes mellitus with sub-optimal blood sugar control, but this was not reported as a complication from the ICD implantation. Results also showed that 60.4% of patients attended the 3-week follow-up and had their cardiac devices interrogated, and out of this proportion, only 48.2% of them attended the 3-month follow-up visit for clinical evaluation and device interrogation. A 6-month follow-up (both in-person and telephone) revealed that 20 (41.7%) of the patients were still alive, some of which reported some clinical improvement; 6 of them (12.5%) were reported to have died within the 6-month post-procedure period; and 22 (45.8%) of them were lost to follow-up.
Heart failure is caused by a structural or functional disorder that negatively affects the pumping function of the heart [14]. It results in significant morbidity and mortality and leads to frequent hospitalizations due to the progressive nature of the condition. People with NYHA class II and III heart failure are more susceptible to sudden cardiac death, and this is the cause of death in 30%-50% of people with heart failure [14]. CRT attempts to improve the synchronized contraction of both the left and right ventricles to improve cardiac output [14], and people with heart failure and wide QRS complexes with a low ejection fraction who are symptomatic despite optimal medical therapy are candidates for CRT implantation [14]. People with a combination of a low ejection fraction and clinical heart failure are at an increased risk of sudden cardiac death from lethal ventricular arrhythmias. They are those who may benefit from CRT treatment with a defibrillator (CRT-D) [14]. The development of biventricular pacing has also proved to reduce the rate of hospitalization and improve patients' quality of life, an important milestone in the management of patients with heart failure [15].
Bradycardia can be caused by conditions that affect the natural pacemakers of the heart, such as sinus node dysfunction (SND) and atrioventricular (AV) blocks. People suffering from bradycardia may present with palpitations, exercise intolerance, and fainting, and the only effective treatment for patients suffering from symptomatic bradycardia is the implantation of a permanent pacemaker [16]. The indications for pacemaker therapy have expanded in the past 45 years, and aside from its use in the treatment of bradyarrhythmias , other indications include the treatment of tachyarrhythmias, certain types of syncope, and advanced heart failure [17]. According to the Canadian Institute for Health Information Cardiac Rate Book 2016, the number of people who received new or replacement implanted cardiac devices annually in Ontario, Canada, for a period of 6 years, 2010-2015, was as follows: pacemaker insertions were between 6,036 and 6,996 people; CRTs without a defibrillator (CRT-Ps) were between 49 and 146 people; and CRT-Ds and ICDs were between 1,798 and 2,208 people [14]. This finding shows that pacemaker implantation was the most common cardiac device therapy done, a finding that is similar to our results, where 20 out of the 48 patients in our study underwent DCPPM insertion, and SND and high-grade AV block were the commonest indications [18]. A report of a study done in a tertiary health institution in Southern Nigeria that described findings from 44 adult patients who had permanent cardiac pacemaker insertion done from June 1st, 2017 to May 31st, 2019, stated that the most common electrocardiography (ECG) finding in 31 of them was complete heart block (70.46%) [19]. In our study, we found the most common indication for DCPPM to be 3rd degree atrioventricular block (50%), and the second most common indication to be SND. SND is a syndrome usually manifested by a variety of cardiac arrhythmias, including sinus bradycardia, sinoatrial block, and paroxysmal supraventricular tachycardia, which frequently alternates with periods of bradycardia, the so-called "bradycardia-tachycardia syndrome".
From the results of our study, 81.2% (39 out of 48 patients) had to pay "out-of-pocket" for their procedure, even if most of them had health insurance, and only 18.7% were privileged to have their health insurance cover their cost. Whereas, in most European countries, the "diagnosis-related group" (DRG) classification is used for patient classification and hospital reimbursement to cover the cost of implanting cardiac electronic devices [20]. And this system ensures that more of the patients who require cardiac device therapy actually get the cost covered by these healthcare plans. This begs the need for a review of health policies or a reduction in the cost of the procedure. There are a couple of complications that can occur with cardiac device therapy, and they include minor complications like pocket hematomas, implantation site infections, slight lead dislodgement, and pneumothorax. These are considered minor because a re-intervention might not be necessary [21]. Major complications could also occur that require re-intervention, and they include severe implantation site infections and systemic infections, e.g. endocarditis, cardiac perforation, permanent malfunction of a pacemaker resulting from manipulation of the pulse generator within its skin pocket (Twiddler syndrome), deep vein thrombosis, stroke, etc. In our study, the complication rate was 6.25% (3 out of 48) and included 1 lead dislodgement and 2 implantation site infections (minor); these were considered minor complications as they did not necessitate re-intervention. Higher complication rates were reported in some studies carried out abroad. One of them was done by Vanegas-Cadavid et al. [22], and involved 897 patients who had cardiac device therapy done in a reference center in Colombia. A complication rate of 10.9% was reported; lead displacement was responsible for 3.6%, and a pocket hematoma was responsible for 3.3%. Seventy-five percent (75%) of these complications were categorized as major complications, and 73.5% occurred in the first month after the procedure. Another was done by Ryan et al. [23], which involved 397 patients who underwent cardiac device therapy in a cardiovascular center in Ireland and reported an overall complication rate of 10.83%. Whether the disparity in complication rate between our center and these centers abroad is a matter of sample size is a matter for consideration.
People with an implanted ICD, CRT, or pacemaker are expected to be followed up for device checks and clinical evaluation. During these visits, the physician, nurse, or technician is able to read the information stored by the device using a magnetically inductive programming wand. This provides information on arrhythmic episodes, delivered therapeutic device responses, and device integrity [14]. This process is called 'device interrogation', and in our cardiovascular facility, it is usually done before the patient is discharged from the hospital, and then every 3 months for the first 6 months, then every 6 months. This protocol is similar to that reported by Guédon-Moreau et al. [24], where the protocol for that center is a clinic visit within 1-3 months from the implantation of the device, then every 6 months. Different 'center-specific protocols' also exist, like that reported by Heidbuchel et al. [25], where clinic visits are done 6 weeks after discharge, then 12 monthly, and that reported by Sardu et al. [26], where clinic visits are done 10 days after the implantation, then at 1, 3, 6, and 12 months.
In our study, 60.4% of the patients attended the 3-week clinic visit for evaluation and cardiac device interrogation, and out of this proportion, 48% of them (about 13 patients) made it to the 3-month follow-up visit. This means that at the 3rd month post-procedure, 35 (72.9%) out of the 48 patients were lost after their cardiac device implantation procedure, together with the information contained in their device. So, in summary, it makes it difficult for the physician to ascertain whether the device is serving its purpose or not or if the patients are experiencing any difficulties with the device. This makes the need for the use of 'remote-monitoring-capable' cardiac devices and the remote interrogation of cardiovascular implantable electronic devices imperative in Nigeria. Remote interrogation evaluation monitoring provides full device interrogation, monitoring for arrhythmias, and cardiovascular implantable electronic device performance surveillance from the patient's home [27]. Data from one study suggested that remote monitoring and evaluation of cardiac devices conferred a 50% relative reduction in the risk of death compared with clinic follow-up [28]. Although our study reported that 12.5% of the patients had died by the 6-month follow-up, excluding those who were lost to follow-up, better survival rates have been reported in much larger studies. One of them was a retrospective study that involved 2108 patients who underwent permanent pacemaker implantation. A total mortality rate of 3.9% (n=83) was recorded, and no deaths were directly related to the procedure. No data was available to indicate the proportion of these deaths that occurred within a year [29]. Another study involved 415 ICD implantations, 80 of which were CRT-Ds, and reported a death rate of 1.4% (n=6) within a year of ICD insertion [30].
The incidence and prevalence of cardiovascular disease are on a steady rise in Nigeria, which means that the number of cases of heart failure and the demand for cardiac device therapy services will increase. Aside from the amount of financial capital required to establish these centers and the scarcity of highly skilled professionals required to perform these procedures, one of the major factors hindering the widespread performance of these procedures in Nigeria is the cost of the procedure. Only a small percentage of the population is able to afford to pay for these procedures 'out-of-pocket', and even a smaller percentage is privileged enough to have the cost covered by their health insurance companies. Among those who are fortunate enough to undergo successful device implantation, subsequent attendance at scheduled follow-up clinic visits for clinical evaluation and cardiac device interrogation is not common. The most likely cause of the poor follow-up is the distance between patients' residences and these few cardiovascular centers scattered all over the country. The solutions to these problems will largely depend on amendments to healthcare and insurance policies, the use of �second-hand' (used) cardiac devices, which are definitely cheaper, and the introduction of remote monitoring-enabled' cardiac devices. Partnerships with foreign non-governmental organizations and major cardiovascular centers and hospitals abroad have been one of the major ways cardiovascular centers in Nigeria have received donations of equipment and supplies for cardiac device therapy at a very affordable rate. Our center has been one of the few beneficiaries of these partnerships and has offered cardiac device therapy to patients at an affordable cost during these periods.
The use of 'remote monitoring enabled' devices and the practice of remote monitoring of implanted cardiac devices in Nigeria will definitely be a 'game-changer' for two major reasons. Firstly, it will ensure that the performance and integrity of the devices implanted in the patients are continuously monitored remotely, and with the information obtained, patients experiencing repeated 'cardioverter-defibrillator shocks', and those with poorly functional devices can be identified early. This information can lead to proactive efforts to contact these patients. Secondly, remote monitoring can encourage the establishment of a 'national cardiac device therapy registry', where details of all patients with a device, together with the details of their device's performance and integrity, are recorded. With this information available and continuous remote monitoring, policies can be created where patients who have passed on but still have high-functioning cardiac devices implanted in them can be identified and located. The policies would enable these high-functioning cardiac devices to be extracted from these dead patients (with the appropriate consent), sterilized adequately, and then implanted in patients who need them at an affordable cost. A functioning system like this will ensure that the cost of procedures will not be a major hindrance to meeting the increasing need for cardiac device therapy in Nigeria.
What is known about this topic
- The increasing prevalence of cardiovascular disease in Nigeria and other developing countries, begs the need for more centers that can offer cardiac device therapy;
- Insertion of a permanent pacemaker is the commonest cardiac device therapy, and atrioventricular blocks and symptomatic bradycardia are the commonest indications.
What this study adds
- The two major problems facing cardiac device therapy in our center were coverage for the cost of the procedure and a poor follow-up system;
- Hence, there is a need for remote monitoring of implanted cardiac devices and the use of second-hand devices to reduce the cost of cardiac device therapy.
The authors declare no competing interests.
Okorie Chinedum Anya, Andrew Kamsoko Ndakotsu and Rupert Ndubisi Chima were involved in the conceptualization and design of the manuscript. Rupert Ndubuisi Chima and Onomhem Leticia Imafidon were involved in data collection, analysis and interpretation. Okorie Chinedum Anya, Eniola Ruth Ajayi and Henry Rotimi Solanke were involved in the initial draft of this manuscript, while Adeyemo Jerry Adesola, Andrew Kamsoko Ndakotsu and Nnamdi Lionel Nwoke handled the proof-reading and editing of this manuscript. All authors have read and approved the final version of this manuscript.
We are grateful to Dr. Ajibola Babatunde and Phys. Elvis Osobaye, managers at Cardiocare Multispecialty Hospital for their cooperation and support.
Table 1: indications for the various cardiac device therapy procedures and their proportions
Figure 1: the individual proportions of the various completed cardiac device therapy procedures
- Grubb N. Device therapy. Journal Royal College of Physicians of Edinburgh. 2006;36(2):136. Google Scholar
- Lak HM, Goyal A. Pacemaker types and selection. 2020. Google Scholar
- DeForge WF. Cardiac pacemakers: a basic review of the history and current technology. Journal of Veterinary Cardiology. 2019;22:40-50. PubMed | Google Scholar
- Moss AJ. Implantable cardioverter defibrillator therapy: the sickest patients benefit the most. Circulation. 2000;101(14):1638-1640. PubMed | Google Scholar
- Wiles BM, Illingworth CA, Paisey JR, Roberts PR, Harden SP. Keeping up appearances: the radiographic evolution of cardiovascular implantable electronic devices. Br J Radiol. 2018 Jan;91(1081):20170506. PubMed | Google Scholar
- Brugada J. Cardiac arrhythmias and sudden death. An article from the e-journal of the ESC Council for Cardiology Practice. 2004;2:32. Google Scholar
- Ghzally Y, Mahajan K. Implantable Defibrillator. 2017. Google Scholar
- Jaffe LM, Morin DP. Cardiac resynchronization therapy: history, present status, and future directions. Ochsner J. 2014 Winter;14(4):596-607. PubMed | Google Scholar
- Cazeau Serge, Ritter Phillipe, Lazarus Arnaud, Gras Daniel, Backdach Hayssam, Mundler Olivier, Mugica Jacques. Multisite pacing for end-stage heart failure: early experience. Pacing Clin Electrophysiol. 1996 Nov;19(11 Pt 2):1748-57. PubMed | Google Scholar
- O'Brien Thomas, Park Myung Soo, Youn Jong Chan, Chung Eugene. The past, present and future of cardiac resynchronization therapy. Korean Circulation Journal. 2019; 49(5):384-399. PubMed | Google Scholar
- Ike SO, Onyema CT. Cardiovascular diseases in Nigeria: What has happened in the past 20 years? Nigerian Journal of Cardiology. 2020 Jan 1;17(1):21-6. Google Scholar
- Bonny A, Ngantcha M, Jeilan M, Okello E, Kaviraj B, Talle MA et al. Statistics on the use of cardiac electronic devices and interventional electrophysiological procedures in Africa from 2011 to 2016: report of the Pan African Society of Cardiology (PASCAR) Cardiac Arrhythmias and Pacing Task Forces. Europace. 2018 Sep 1;20(9):1513-1526. PubMed | Google Scholar
- Vogt J, Heintze J, Hansky B, Güldner H, Buschler H, Horstkotte D. Implantation: tips and tricks-the cardiologist's view. European Heart Journal Supplements. 2004; 6(suppl_D):D47-D52. Google Scholar
- Health Quality Ontario. Remote monitoring of implantable cardioverter-defibrillators, cardiac resynchronization therapy and permanent pacemakers: a health technology assessment. Ont Health Technol Assess Ser. 2018 Oct 24;18(7):1-199. PubMed | Google Scholar
- Ganjehei L, Razavi M, Massumi A. Cardiac resynchronization therapy: a decade of experience and the dilemma of nonresponders. Texas Heart Institute Journal. 2011; 38(4):358. PubMed | Google Scholar
- Edwards SJ, Karner C, Trevor N, Wakefield V, Salih F. Dual-chamber pacemakers for treating symptomatic bradycardia due to sick sinus syndrome without atrioventricular block: a systematic review and economic evaluation. Health Technology Assessment (Winchester, England). 2015;19(65):1-210. PubMed | Google Scholar
- Gregoratos G. Indications and recommendations for pacemaker therapy. Am Fam Physician. 2005 Apr 15;71(8):1563-70. PubMed | Google Scholar
- Dalia T, Amr BS. Pacemaker Indications. Europe PMC. 2018. PubMed | Google Scholar
- Edafe Emmanuel, Odia James, Akpa Maclean, Dodiyi-Manual Sotonye, Oye Herbert, Agadah Zuoboemi. Experience with cardiac pacing in the Nigerian Delta region. Nigerian Journal of Cardiology. 2020; 17(2):136. Google Scholar
- Boriani G, Burri H, Mantovani LG, Maniadakis N, Leyva F, Kautzner J et al. Device therapy and hospital reimbursement practices across European countries: a heterogeneous scenario. Europace. 2011;13(suppl_2):ii59-65. PubMed | Google Scholar
- Kirkfeldt RE, Johansen JB, Nohr EA, Jørgensen OD, Nielsen JC. Complications after cardiac implantable electronic device implantations: an analysis of a complete, nationwide cohort in Denmark. Eur Heart J. 2014 May;35(18):1186-94. PubMed | Google Scholar
- Vanegas-Cadavid DI, Ibatá-Bernal L, Franco-Garrido PA, Valderrama-Barbosa Z. Complications related to cardiac implantable electronic devices. Revista Colombiana de Cardiología. 2020;27(5):420-427. Google Scholar
- Ryan N, Mahon N, Walsh K, O'Neill J, Keelan E, Keelan P et al. Cardiac implantable electronic devices complications�a single centre experience. Heart. 2015; 101(Suppl 5):A6-A7. PubMed | Google Scholar
- Guédon-Moreau L, Kouakam C, Klug D, Marquié C, Brigadeau F, Boulé S et al. Decreased delivery of inappropriate shocks achieved by remote monitoring of ICD: a substudy of the ECOST trial. J Cardiovasc Electrophysiol. 2014 Jul;25(7):763-70. PubMed | Google Scholar
- Heidbuchel H, Hindricks G, Broadhurst P, Van Erven L, Fernandez-Lozano I, Rivero-Ayerza M et al. EuroEco (European Health Economic Trial on Home Monitoring in ICD Patients): a provider perspective in five European countries on costs and net financial impact of follow-up with or without remote monitoring. European Heart Journal. 2015;36(3):158-169. PubMed | Google Scholar
- Sardu C, Santamaria M, Rizzo MR, Barbieri M, di Marino M, Paolisso G et al. Telemonitoring in heart failure patients treated by cardiac resynchronisation therapy with defibrillator (CRT-D): the TELECART Study. Int J Clin Pract. 2016 Jul;70(7):569-76. PubMed | Google Scholar
- Cronin EM, Ching EA, Varma N, Martin DO, Wilkoff BL, Lindsay BD. Remote monitoring of cardiovascular devices: a time and activity analysis. Heart Rhythm. 2012 Dec;9(12):1947-51. PubMed | Google Scholar
- Heidbüchel Hein, Lioen P, Foulon S, Huybrechts W, Ector J, Willems R et al. Potential role of remote monitoring for scheduled and unscheduled evaluations of patients with an implantable defibrillator. Europace. 2008 Mar;10(3):351-7. PubMed | Google Scholar
- Villalba S, Roda J, Quesada A, Palanca V, Zaragoza C, Bataller E et al. Retrospective study of patients who undergo pacemaker implantation in short-stay ambulatory surgery: Long-term follow-up and cost analysis. Rev Esp Cardiol. 2004 Mar;57(3):234-40. PubMed | Google Scholar
- Wheen P, Sharif Z, O'Carroll-Lolait C, Moore D. 42 A review of mortality in patients who undergo ICD insertion in a single implantation centre. Heart. 2020;106(Suppl 4):A27-A28. PubMed | Google Scholar