Oral hygiene and periodontal treatment interventions for reducing blood pressure in hypertensive adults from low and middle-income countries: a systematic review
Agnes Gatarayiha, Caleb Kimutai Sagam, Joseph Ntaganira, Stephen Rulisa, Léon Mutesa, Zoe Brookes
Corresponding author: Agnes Gatarayiha, School of Dentistry, College of Medicine and Health Sciences, University of Rwanda, Kigali, Rwanda 
Received: 26 Jan 2024 - Accepted: 05 Mar 2024 - Published: 11 Jun 2024
Domain: Dental Public Health,Periodontics,Non-Communicable diseases epidemiology
Keywords: Oral hygiene, periodontal therapy, high blood pressure, cardiovascular health, hypertensive patients
©Agnes Gatarayiha et al. PAMJ Clinical Medicine (ISSN: 2707-2797). This is an Open Access article distributed under the terms of the Creative Commons Attribution International 4.0 License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Cite this article: Agnes Gatarayiha et al. Oral hygiene and periodontal treatment interventions for reducing blood pressure in hypertensive adults from low and middle-income countries: a systematic review. PAMJ Clinical Medicine. 2024;15:20. [doi: 10.11604/pamj-cm.2024.15.20.42793]
Available online at: https://www.clinical-medicine.panafrican-med-journal.com//content/article/15/20/full
Review 
Oral hygiene and periodontal treatment interventions for reducing blood pressure in hypertensive adults from low and middle-income countries: a systematic review
Oral hygiene and periodontal treatment interventions for reducing blood pressure in hypertensive adults from low and middle-income countries: a systematic review
![]() Agnes Gatarayiha1,&, Caleb Kimutai Sagam2,
Agnes Gatarayiha1,&, Caleb Kimutai Sagam2, ![]() Joseph Ntaganira3, Stephen Rulisa4,
Joseph Ntaganira3, Stephen Rulisa4, ![]() Léon Mutesa5, Zoe Brookes6
Léon Mutesa5, Zoe Brookes6
&Corresponding author
Introduction: while previous systematic reviews have established a link between periodontal diseases and hypertension in high-income countries, evidence from Low and Middle-Income Countries (LMICs) remains scarce. This systematic review aimed to fill this gap by evaluating the impact of oral hygiene education and periodontal therapy on reducing high blood pressure in hypertensive patients.
Methods: we conducted a comprehensive search of clinical trials and cohort studies published between 2000-2022 across PubMed, SCOPUS, Medline, CINAHL, and Google Scholar. Secondary outcomes included changes in blood levels in pro-inflammatory C-reactive protein (CRP) and interleukin-6 (IL-6), and anti-inflammatory interleukin-10 (IL-10). Quality and risk of bias were evaluated using the Joanna Brings Institute Appraisal Checklists.
Results: out of 519 records, we included four articles that met the eligibility criteria. Three studies from Brazil and one from Syria assessed the primary outcomes of systolic and diastolic blood pressure (SBP/DBP) among hypertensive patients (SBP/DBP≥140/90mmHg) undergoing periodontal treatment and oral hygiene education. Three out of four studies demonstrated important reductions in SBP and DBP with average reductions of 12.5 mmHg and 10.0 mmHg, respectively, in response to oral hygiene and periodontal therapy. Furthermore, three studies reported decreases in systemic inflammatory markers, including CRP, cytokines, triglycerides, and erythrocyte sedimentation rate (ESR).
Conclusion: this systematic review underscores the potential of periodontal therapy and oral hygiene education in managing hypertension, especially in resource-constrained LMICs. Further research is essential to validate these findings and address the specific needs of diverse populations in these regions.
Hypertension is globally defined as a systolic arterial blood pressure (SBP) ≥ 140mmHg and a diastolic blood pressure (DBP) ≥90mmHg [1], affecting over 30% of the adult population worldwide. It stands as a leading causative factor for cardiovascular diseases [2], resulting in high mortality rates due to fatal heart attacks and strokes [3]. Confirmed risk factors for hypertension, include poor oral hygiene, diabetes, and tobacco smoking [4].
Periodontitis is a major global health burden, with an estimated 50% of the population affected globally and severe periodontitis represents the 6th most prevalent disease worldwide (11.2%) [5,6]. Despite variations in periodontal disease classifications globally, severe periodontitis remains a significant concern [7,8]. In the USA, the National Health and Nutrition Examination Survey (NHANES) between 2009-2012 revealed that 8.9% of the population presented with severe periodontitis [9]. In Low and Middle-Income Countries (LMICs), as defined as countries with low, lower-middle, or upper-middle-income economy according to the World Economic Situation and Prospect (WESP) and World Bank [10,11], the prevalence of severe periodontitis tends to be much higher, such as 42.2% in Ethiopia [12] and 43.4% in the Ivory Coast [13]. Prevalence of periodontitis was also high in East African countries, with Rwanda reporting 39.6% of their adult population as having periodontitis among people attending the Rwanda Military Hospital [14]. In Uganda, 28.6% of young adults [15], and in Kenya varies between 12-24% in adults [16]. These disparities highlight the need for intervention studies on oral and systemic health in specific LMICs populations.
Recent studies have also shown that oral disease including periodontitis (gum disease), may also be associated with hypertension. However, data on these associated risks are mainly derived from high income countries, emphasizing the need for more mechanistic and interventional studies in low and middle-income countries [5,17,18]. Elevated cardiovascular markers such as CRP, IL-6, fibrinogen, and systemic inflammation are predictors of increased blood pressure [19,20], making them potential secondary outcomes, to assess systemic responses to both hypertension and periodontitis, aspects not fully evaluated in previous systematic reviews.
Individuals with moderate to severe periodontitis appear to have greater (20%) odds of developing hypertension [21], with the likelihood increasing with hypertension (49%) [9]. In addition, Desvarieux et al. demonstrated in the USA that people with periodontitis had a higher risk of developing hypertension, associated with an increasing bacterial load due to subgingival plaque [22]. Systematic reviews from developed countries including Poland, Japan, Sweden, and USA, consistently demonstrate two to three times higher odds for hypertension in individuals with periodontitis [23,24]. Studies from Tanzania, India, and Cameroun have reported an association between periodontitis and hypertension [25,26]. Therefore, there is need to study both hypertension and periodontitis considering the scarcity of evidence in Africa, (LMICs) [27].
Another pertinent piece of evidence is the observation that successful treatment of periodontal disease by non-surgical therapy (sometimes called root planning, root surface debridement depending on the country) may lower blood pressure, namely decrease hypertension [24,28]. Hence, this systematic review aims to evaluate oral hygiene education and periodontal therapy's effectiveness in reducing high blood pressure among hypertensive patients in LMICs.
Protocol and registration
This review followed the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines [29]. The systematic review protocol was registered in PROSPERO with registration ID: CRD42022375002.
Search strategy
We searched the following electronic databases: PubMed, Medline, CINAHL EBSCO HOST Scopus,, with a supplement of Google Scholar for relevant published articles in English from January 2000 to December 2022. We used the search terms to identify relevant articles: periodontal diseases, periodontitis, periodontic, periodontal attachment loss, hypertension, high blood pressure, arterial blood pressure, oral hygiene, oral hygiene instruction, periodontal treatment, dental scaling, dental prophylaxis, periodontal therapy, sub gingival scaling, root planning. We used Boolean operators such as "AND" and "OR" to find out the related articles. For additional articles, we conducted a manual search and examined reference lists to retrieve relevant citations.
Study eligibility
We followed PICO outline for this review. We included studies on hypertensive and pre-hypertensive individuals (female and males) with periodontitis who were aged from 18 to 65 years old. We also included studies that evaluated the efficacy of Oral Hygiene Education and periodontal therapy (supra and subgingival scaling) by comparing them with the usual care. The outcomes were reduction of diastolic and Systolic Blood Pressure (SBP) following periodontal therapy and oral hygiene education. Furthermore, primary outcomes were changes in blood pressure (SBP and DBP) among patients undergoing periodontal treatment and oral hygiene instructions, whereas the secondary outcomes were changes in systemic levels of CRP and pro-inflammatory (IL-6,) and anti-inflammatory (IL-10) cytokines, endothelial function, oral bacteria and periodontal clinical parameters of hypertensive patients such as clinical attachment loss, pocket depths and bleeding on probing.
Inclusion and exclusion criteria
This study considered only articles published in peer-reviewed journals with the following inclusion criteria: randomized clinical trials, cohort studies, and case-control studies. Additionally, reported articles in English focusing on periodontitis (all stages), oral hygiene and periodontal treatment (non-surgical therapy), and reducing high blood pressure defined as hypertension according to the appropriate classification system were included. Articles on case reports, case series, cross-sectional studies, and those reporting on pregnant and lactating women with hypertension were excluded, as well as articles on other systemic diseases other than hypertension. Furthermore, articles reporting on countries other than Low and Middle-Income Countries (LMICs) as defined by the World Economic Situation and Prospect (WESP) and World Bank [10,11] were excluded.
Screening and selection of papers
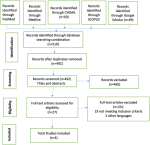
This process involved two steps. Firstly, the retrieved titles and abstracts detected by the search were exported to covidence software [30], duplicates were removed. Secondly, two independent reviewers independently screened the titles and abstracts and conducted full-text assessments of potential eligible studies. Any disagreements were resolved by consensus through discussion if not, a third reviewer was consulted. A list of excluded studies is provided in Annex 1. Two reviewers who retrieved the potential eligible studies included in our study performed independent full-text reading. Four included studies (two cohorts and two clinical trials) that met the inclusion were captured. The results of the search are presented in a PRISMA flow diagram (Figure 1).
Data extraction
Two authors and an assistant researcher independently performed data extraction. The included studies were presented in an evidence table describing study characteristics, population, type of interventions, and effect of intervention on periodontal diseases and high blood pressure (hypertension). Any disagreements that arose between the reviewers were resolved through discussion. Characteristics of included studies are presented in Table 1.
Synthesis of the results
Due to the high degree of heterogeneity in the included studies (study designs, intervention details, outcome measures), meta-analysis was not conducted. We presented our results in tables and synthesized our results narratively.
A flow diagram illustrating the studies selection process is presented in Figure 1. The electronic searches across five databases yielded 519 records, which were then reduced to 492 records after removing duplicates. A total of 492 records were screened for titles and abstracts. Subsequently, 27 articles underwent full-text screening, resulting in the exclusion of 23 articles that did not meet the eligible criteria outlined in our inclusion criteria. The excluded studies are detailed in Annex 1. Finally, four articles were included in this review.
Study characteristics
All included studies are summarized in Table 1. Among the four included studies, two were randomized clinical trials, and two were cohort studies. The sample sizes ranged from 22 to 64 participants [31,32].
Risk of bias assessment
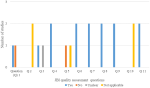
The methodological quality utilized the Joanna Briggs Institute appraisal checklist for the critical appraisal of the included studies [33]. All included studies were appraised for quality by two independent reviewers, with contentious issues resolved through discussion. Randomized controlled trials (RCTs) generally reported most items compared to cohort studies. The Figure 2 and Figure 3 presents the methodological quality assessment of cohort studies and RCTs, respectively. The PRISMA guidelines were followed, and any discrepancies were resolved through discussions.
Confirmation of an effective intervention
Confirmation of successful interventions (oral hygiene and periodontal therapy) is crucial for the validity of this review, considering the known variability in managing periodontal disease among clinicians. The interventions varied, with one study involving non-surgical periodontal therapy only [34] and the other three studies involving a combination of oral hygiene instruction and non-surgical therapy [31,32,35].
The oral hygiene education in both cohort studies involved demonstrating tooth brushing techniques and utilization of dental floss. The successful intervention was confirmed by decreases in plaque and bleeding on probing, from 45.7% to 19.4% after intervention [34,35]. These studies reported improvement in periodontal outcomes, including periodontal Pocket Depth (PD), (4 to 5mm in 19.5%-4.9% sites, P<0.001 and > 6mm in 9.1%-1.2% of sites, P<0.02) and Clinical Attachment Loss (CAL), (4 to 5mm in 23.7%-18.4% of sites, P<0.014; CAL≥6 in 24.5% -11.2% of sites, P<0.002). Notably in the control group, there was no change in periodontal parameters at baseline at 3 months follow up to the oral hygiene and non-surgical therapy intervention. Furthermore, Caula et al. (2014) demonstrated additional improvements in periodontal pocket depth, bleeding on probing, clinical attachment loss, and enhanced indices of oral hygiene at 6 months following the intervention [32].
Periodontal therapy in three studies involved supra and sub-gingival scaling using both manual and ultrasonic machines, alongside root planning to treat periodontal pocket depths greater than 4mm (PDD>4mm). Despite some variation in approach, successful treatment was confirmed by significant (p<0.05) reduction in pocket depths, bleeding on probing and clinical attachment loss at 6 weeks to 3 months [31,32,34].
Primary study outcomes
The intervention led to an immediate decrease in blood pressure after 6 weeks to 3 months in hypertensive patients after treatment [31,34]. Significant decreases in both mean systolic blood pressure, SBP (175 to 157 mmHg) and mean diastolic blood pressure, DBP (from 105 to 95 mmHg) were observed in response to oral hygiene and periodontal therapy.
Confirmation of successful interventions (oral hygiene and periodontal therapy) is essential for the validity of this review, bearing in mind the known variability in the successful management of periodontal disease amongst clinicians [36]. For non-surgical therapy, the intervention in two cohort studies involved supra and sub gingival scaling with both manual and ultrasonic machines along with root planning in periodontal pocket depths of greater than 4mm (PDD>4mm). This resulted in reduced pocket depths, bleeding on probing and clinical attachment loss, all indicative of a reduced severity of periodontal diseases, confirming the successful treatment.
Non-surgical therapy and oral hygiene education intervention in both cohort studies also included a demonstration of tooth brushing techniques and the utilization of dental floss. The success of this intervention was confirmed by decreases in plaque and bleeding on probing, from 45.7% to 19.4% after the intervention. At 6 months follow-up after non-surgical therapy and oral hygiene intervention, blood pressure and periodontal clinical parameters still demonstrated a significant association between successful periodontal treatment and the reduction of blood pressure during hypertension among study patients, with treatment provided alongside oral health education [31,32].
Secondary outcomes
Hypertensive patients undergoing concurrent periodontal treatment and oral hygiene education concurrently reported a positive impact on reduction of SBP (175 to 157 mmHg) and DBP (105 to 95 mmHg). They also experienced a significant decrease in cardiovascular biomarkers, including fibrinogen (from 376gm/dl to 344gm/dl, p<0.001) CRP (from 0.6mg/dl to 0.3mg/dl; p<0.001) and IL-6 (from 2.1pg/ml to0.9pg/ml) [34,35].
From the two clinical trials included in this systemic review involving hypertensive patients, significant changes were observed at baseline and 3 months follow up after the intervention. One study demonstrated a notable decrease in bleeding on probing (BOP) (45.5%-11.4% of sites; P<0.002), pocket depth (PD) (4 to 5mm in 19.5%-4.9% sites, P<0.001 and > 6mm in 9.1%-1.2% of sites, P<0.02) and CAL (4 to 5mm in 23.7%-18.4% of sites, P<0.014; CAL>or=6 in 24.5% -11.2% of sites, P<0.002). In contrast, the control group showed no change in periodontal parameters. Additionally, a significant reduction in cardiovascular markers was noted in hypertensive patients following the intervention, including IL-6 (24.4-10pg/ml, P=0.03), fibrinogen (407-348mg/dl, P=0.003) and CRP (1.4-0.9mg/dl, P=0.005), all associated with reduced blood pressure, at baseline and 3 months follow up. Conversely, in the control group, levels of CRP (0.5-1.1mg/dl, P=0.01) and IL-6 (7-24.9mg/dl, P=0.01) increased significantly [31]. The two clinical trials included in this review reported a reduction in cardiovascular markers after 3-6 months of follow-up, along with improvement in Systolic Arterial Pressure (SAP) and Diastolic Arterial Pressure (DAP) in hypertensive patients [32].
The current systematic review aimed to evaluate the effectiveness of oral hygiene and periodontal treatment interventions (non-surgical periodontal therapy) in reducing high blood pressure among adults with hypertension in Low and Middle-Income Countries (LMICs). The interventions demonstrated concurrent effectiveness in improving periodontal disease indicators, such as bleeding on probing, pocket depths, and clinical attachment loss among hypertensive individuals. The findings of this systematic review align with previous evidence from high-income countries, emphasizing the association between periodontal diseases and hypertension [37-40]. Secondary outcome measures indicated that periodontitis in LMICs was associated with elevated cardiovascular markers, including CRP, IL-6, and fibrinogen, which are predictors of high blood pressure [41,42].
This systematic review affirms the impact of periodontal treatment combined with oral hygiene instruction in reducing blood pressure among hypertensive patients in LMICs. Four included studies support the relationship, in turn supporting the association between periodontal disease and hypertension. The association between periodontal diseases and hypertension is consistent with other studies predicting the gingival bleeding up and probing pocket depth correlating with hypertension in high-income countries [43]. However, the geographical distribution of the included studies, with one cohort study from Syria, and three (two clinical trials and one cohort study) from Brazil, underscores a critical gap in research conducted in Africa, particularly sub-Saharan Africa. This highlights the urgent need for intervention studies addressing the relationship between periodontal diseases and hypertension, in a broader range of geographic locations, especially in Africa.
Regarding follow-up durations, cohort studies from Syria and Brazil reported no reduction in blood pressure among hypertensive patients at 6 to 12 months post non-surgical periodontal treatment together with oral hygiene education. These findings contrast with other studies showing a decrease in blood pressure immediately following interventions, emphasizing the importance of sustained oral hygiene practices for long-term effectiveness [3,5,36,44]. Similarly, in previous studies, there was no decrease in blood pressure within a long-term clinical trial after non-surgical periodontal treatment including oral hygiene education [44,45]. These findings may be related to the fact that excellent personal oral hygiene is key to long-term periodontal treatment effectiveness, yet many patients fail to maintain immaculate oral hygiene. Effective oral hygiene and periodontal therapy were demonstrated in two clinical trials, as evidenced by improved periodontal parameters in the intervention group compared to the control group. These improvements included a significant decrease in bleeding on probing, pocket depth, and clinical attachment loss.
The review also delves into the impact of periodontal therapy on systemic inflammatory biomarkers, such as CRP, IL-6, and fibrinogen, which are elevated in periodontitis patients. The observed reductions in these biomarkers following periodontal treatment and oral hygiene education align with the broader literature associating improved oral hygiene with decreased inflammation and cardiovascular risk [46]. One explanation for increased levels of inflammatory mediators following interventions is that periodontal pathogens reach the bloodstream through periodontal pockets, then initiating inflammatory responses involving elevated inflammatory biomarkers, such as CRP, TNF-α, and IL-6 [3,41,47,48]. Indeed, increases in these systemic inflammatory biomarkers are linked to poor oral hygiene and other cardiovascular diseases, therefore improvement of oral hygiene has a direct effect on the reduction of inflammatory biomarkers [3,48]. Similarly, in the studies reviewed here, periodontal treatment and oral hygiene induced significant reductions in cardiovascular markers, such as IL-6 (P=0.03), fibrinogen (P=0.003), CRP (P=0.005).
Furthermore, if oral hygiene education and periodontal therapy lead to a reduction in cardiovascular biomarkers, as shown in the studies included in this review, this evidence may support a causal explanation for the link between periodontal disease and hypertension. Changes in cardiovascular inflammatory markers in individuals with periodontitis can also contribute to vascular alterations and arterial stiffness, culminating in hypertension [49]. Prolonged vascular alterations may, in turn, progress to atherosclerosis, emphasizing the potential long-term implications of periodontal diseases [41]. The impact of periodontal diseases on systemic vasculature is multifaceted, involving decreased release of endotoxins from oral bacteria. This process, initiates CRP and anti-inflammatory cytokines, leading to endothelial dysfunction [36,50].
The significance of this systematic review extends beyond specific regions or income categories. It holds relevance for scientists and individuals worldwide, reflecting the emergence of global communities with increasingly diverse populations. Despite its valuable insights, the limited number of studies included in this review necessitates caution in drawing conclusive links between periodontitis and hypertension in countries beyond Syria and Brazil. The critical dearth of research in Africa, particularly in sub-Saharan Africa, underscores an urgent need for comprehensive studies due to the high prevalence of periodontitis in these populations. The scarcity of African studies during the literature search resulted in none being included in this systematic review, emphasizing the pressing requirement for research initiatives that can contribute to a more comprehensive understanding of the global implications of the relationship between periodontitis and hypertension.
Moreover, the limited recognition of blood pressure management as a strategy to improve oral health in hypertensive patients, in contrast to diabetes, where the links between periodontal disease and poor glycemia control have been more widely proven [51,52] underscores the need for increasing awareness and integration of oral health practices in hypertension management guidelines.
The study recommends frequent tooth brushing, effective interdental cleaning, and regular dental service visits for patients with periodontitis particularly those with elevated blood pressure [48]. By reducing oral and then systemic inflammation, oral health promotion may potentially decrease the risk and progression of cardiovascular disease. The four studies in this review suggest that preventing and treating periodontitis may reduce the risk of occurrence of hypertension in certain LMICs, similar to high-income countries where systematic review research has reported a similar association [24,45,47,53].
Limitation
Despite its contributions, this systematic review faces limitations. The scarcity of studies in Africa precluded a comprehensive analysis of the link between periodontitis and hypertension in this region. Methodological variations among the included studies, including population differences, study settings, and diagnostic criteria for periodontitis, further complicate cross-country comparisons. The absence of intraoral radiographic examinations in all studies raises concerns about the accuracy of periodontal disease diagnosis.
In conclusion, this research contributes to the global understanding of the role of oral health in hypertensive patients, advocating for the integration of oral hygiene and periodontal non-surgical therapy in hypertension management in LMICs. The study highlights the potential of periodontal therapy to reduce the risk and progression of cardiovascular diseases by addressing oral and systemic inflammation. The findings underscore the importance of oral health promotion in reducing the occurrence of hypertension, mirroring associations observed in high-income countries. Recommendations for LMICs include prioritizing oral health practices alongside conventional hypertension management strategies.
What is known about this topic
- Association between periodontal diseases and hypertension has been consistently reported in high-income settings;
- People with periodontitis are at higher risk of developing hypertension from increased bacterial load due to sub-gingival plaques.
What this study adds
- This study highlights the potential of integrating oral hygiene education and periodontal therapy in the management of hypertension;
- Additionally, it also emphasizes on increase oral health knowledge and practices among hypertensive patients.
The authors declare no competing interests.
All authors contributed to the conception, design, and interpretation of results. Agnes Gatarayiha participated in the writing of the first draft. All authors revised critically the manuscript and agreed to submit the final manuscript for publication. All authors read and approved the final manuscript.
We would like to thank Thierry Uhawenimana for his input in methodological guidance. We thank the research assistants Donat Uwayezu and Usiel Nsabimana for their assistance in screening titles and abstracts to data extraction inputs. We acknowledge the training on conducting systematic review and meta-analysis funded by the collaboration for Evidence-based Healthcare and Public Health in Africa (CEBHA+) and also acknowledge the support from the Capacity Building for Female Scientists in East Africa program (CaFe-SEA) under the East African Consortium for Clinical Research (EACCR) partner's institution funded by European and Developing Countries Clinical Trials Partnership (EDCTP).
Table 1: characteristics and main findings of included studies
Figure 1: PRISMA flow diagram/chart illustrating the study selection process
Figure 2: quality assessment of cohort studies included in the review
Figure 3: quality assessment for RCTs included in the review
Annex 1: list of excluded studies (PDF - 113KB)
- WHO. WHO Technical Report Series 862, hypertension control. 2015. World Health Organisation. Geneva. 1996. Accessed 20th November 2022.
- Pietropaoli D, Del Pinto R, Ferri C, Wright JT Jr, Giannoni M, Ortu E et al. Poor Oral Health and Blood Pressure Control Among US Hypertensive Adults. Hypertension. 2018 Dec;72(6):1365-1373. PubMed | Google Scholar
- Sharma S, Sridhar S, McIntosh A, Messow CM, Aguilera EM, Del Pinto R et al. Periodontal therapy and treatment of hypertension-alternative to the pharmacological approach. A systematic review and meta-analysis. Pharmacol Res. 2021 Apr;166:105511. PubMed | Google Scholar
- Sanz M, D'aiuto F, Deanfield J, Fernandez-Avil's F. European workshop in periodontal health and cardiovascular disease - Scientific evidence on the association between periodontal and cardiovascular diseases: A review of the literature. Eur Hear Journal Suppl. April 2010;12(suppl_B):B3-B12. Google Scholar
- Machado V, Aguilera EM, Botelho J, Hussain SB, Leira Y, Proença L et al. Association between Periodontitis and High Blood Pressure: Results from the Study of Periodontal Health in Almada-Seixal (SoPHiAS). J Clin Med. 2020 May 23;9(5):1585. PubMed | Google Scholar
- Tonetti MS, Jepsen S, Jin L, Otomo-Corgel J. Impact of the global burden of periodontal diseases on health, nutrition and wellbeing of mankind: A call for global action. J Clin Periodontol. 2017 May;44(5):456-462. PubMed | Google Scholar
- Oh SL, Yang JS, Kim YJ. Discrepancies in periodontitis classification among dental practitioners with different educational backgrounds. BMC Oral Health. 2021 Jan 22;21(1):39. PubMed | Google Scholar
- Papapanou PN, Sanz M, Buduneli N, Dietrich T, Feres M, Fine DH et al. Periodontitis: Consensus report of workgroup 2 of the 2017 World Workshop on the Classification of Periodontal and Peri-Implant Diseases and Conditions. J Periodontol. 2018 Jun;89 Suppl 1:S173-S182. PubMed | Google Scholar
- Eke PI, Dye BA, Wei L, Slade GD, Thornton-Evans GO, Borgnakke WS et al. Update on Prevalence of Periodontitis in Adults in the United States: NHANES 2009 to 2012. J Periodontol. 2015 May;86(5):611-22. PubMed | Google Scholar
- United Nations. World Economic Situation and Prospects 2023. Department of Economic and Social Affairs Publications. 2023. Accessed 20th November 2022.
- World Bank. World Bank classification of country economies (June 2023). World Bank. 2023. Accessed 20th November 2022.
- Tefera A, Bekele B. Periodontal Disease Status and Associated Risk Factors in Patients Attending a Tertiary Hospital in Northwest Ethiopia. Clin Cosmet Investig Dent. 2020 Nov 10;12:485-492. PubMed | Google Scholar
- Koffi-Coulibaly NT, Pockpa ZAD, Mobio GS, Struillou X, Soueidan A. Prevalence and risk indicators for severe periodontitis in Côte d'Ivoire. J Adv Periodontol Implant Dent. 2022 May 31;14(1):7-12. PubMed | Google Scholar
- Mutamuliza J, Rwema F, Rulisa S, Ntaganira J. Prevalence and Associated Risk Factors of Periodontal Disease among Adults Attending Dental Department in Rwanda Military Hospital (Rwanda): A Cross Sectional Study. Dent Open J. 2015;2(4):105-111. Google Scholar
- Wandera M, Twa-Twa J. Baseline survey of oral health of primary and secondary school pupils in Uganda. Afr Health Sci. 2003 Apr;3(1):19-22. PubMed | Google Scholar
- Oyaro B, Lokken E, Alumera H, Hussein S, Richardson B, Mandaliya K et al. Prevalence and correlates of periodontitis among Kenyan women planning to conceive. BMC Oral Health. 2022 May 31;22(1):216. PubMed | Google Scholar
- El Bcheraoui C, Memish ZA, Tuffaha M, Daoud F, Robinson M, Jaber S et al. Hypertension and its associated risk factors in the kingdom of saudi arabia, 2013: a national survey. Int J Hypertens. 2014;2014:564679. PubMed | Google Scholar
- Bijani M, Parvizi S, Dehghan A, Sedigh-Rahimabadi M, Rostami-Chijan M, Kazemi M et al. Investigating the prevalence of hypertension and its associated risk factors in a population-based study: Fasa PERSIAN COHORT data. BMC Cardiovasc Disord. 2020 Dec 1;20(1):503. PubMed | Google Scholar
- Sanz M, Marco Del Castillo A, Jepsen S, Gonzalez-Juanatey JR, D'Aiuto F, Bouchard P et al. Periodontitis and cardiovascular diseases: Consensus report. J Clin Periodontol. 2020 Mar;47(3):268-288. PubMed | Google Scholar
- Zardawi F, Gul S, Abdulkareem A, Sha A, Yates J. Association Between Periodontal Disease and Atherosclerotic Cardiovascular Diseases: Revisited. Front Cardiovasc Med. 2021 Jan 15;7:625579. PubMed | Google Scholar
- Muñoz Aguilera E, Leira Y, Miró Catalina Q, Orlandi M, Czesnikiewicz-Guzik M, Guzik TJ et al. Is systemic inflammation a missing link between periodontitis and hypertension? Results from two large population-based surveys. J Intern Med. 2021;289(4):532-546. PubMed | Google Scholar
- Desvarieux M, Demmer RT, Jacobs DR Jr, Rundek T, Boden-Albala B, Sacco RL et al. Periodontal bacteria and hypertension: the oral infections and vascular disease epidemiology study (INVEST). J Hypertens. 2010 Jul;28(7):1413-21. PubMed | Google Scholar
- Inoue K, Kobayashi Y, Hanamura H, Toyokawa S. Association of periodontitis with increased white blood cell count and blood pressure. Blood Press. 2005;14(1):53-8. PubMed | Google Scholar
- Martin-Cabezas R, Seelam N, Petit C, Agossa K, Gaertner S, Tenenbaum H et al. Association between periodontitis and arterial hypertension: A systematic review and meta-analysis. Am Heart J. 2016 Oct;180:98-112. PubMed | Google Scholar
- Yamori M, Njelekela M, Mtabaji J, Yamori Y, Bessho K. Hypertension, periodontal disease, and potassium intake in nonsmoking, nondrinker african women on no medication. Int J Hypertens. 2011;2011:695719. PubMed | Google Scholar
- Ghosh S, Kumar M. Prevalence and associated risk factors of hypertension among persons aged 15-49 in India: a cross-sectional study. BMJ Open. 2019 Dec 16;9(12):e029714. PubMed
- Belinga LEE, Ngan WB, Lemougoum D, Nlo'o ASPE, Bongue B, Ngono A et al. Association between periodontal diseases and cardiovascular diseases in Cameroon. J Public Health Afr. 2018 Jul 6;9(1):761. PubMed | Google Scholar
- Luo Y, Ye H, Liu W, Lv Z, Jia Y, Li C et al. Effect of periodontal treatments on blood pressure. Cochrane Database Syst Rev. 2021 Dec 12;12(12):CD009409. PubMed | Google Scholar
- Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021 Mar 29;372:n71. PubMed | Google Scholar
- Covidence. Covidence systematic review software, veritas health innovation. Accessed 20th November 2023.
- Vidal F, Figueredo CM, Cordovil I, Fischer RG. Periodontal therapy reduces plasma levels of interleukin-6, C-reactive protein, and fibrinogen in patients with severe periodontitis and refractory arterial hypertension. J Periodontol. 2009 May;80(5):786-91. PubMed | Google Scholar
- Caúla AL, Lira-Junior R, Tinoco EM, Fischer RG. The effect of periodontal therapy on cardiovascular risk markers: a 6-month randomized clinical trial. J Clin Periodontol. 2014 Sep;41(9):875-82. PubMed | Google Scholar
- Aromataris E Godfrey C, Holly C, Kahlil H, Tungpunkom P. Checklist for Systematic Reviews and Research Syntheses. Joanna Briggs Inst. 2017. Accessed 26 January 2024.
- Albush MM, Razan KK, Raed AD. Effect of surgical and non-surgical periodontal debridement on vascular thrombotic markers in hypertensives. J Indian Soc Periodontol. 2013 May;17(3):324-9. PubMed | Google Scholar
- Vidal F, Cordovil I, Figueredo CM, Fischer RG. Non-surgical periodontal treatment reduces cardiovascular risk in refractory hypertensive patients: a pilot study. J Clin Periodontol. 2013 Jul;40(7):681-7. PubMed | Google Scholar
- Zhou QB, Xia WH, Ren J, Yu BB, Tong XZ, Chen YB et al. Effect of Intensive Periodontal Therapy on Blood Pressure and Endothelial Microparticles in Patients With Prehypertension and Periodontitis: A Randomized Controlled Trial. J Periodontol. 2017 Aug;88(8):711-722. PubMed | Google Scholar
- Muñoz Aguilera E, Suvan J, Buti J, Czesnikiewicz-Guzik M, Barbosa Ribeiro A, Orlandi M et al. Periodontitis is associated with hypertension: a systematic review and meta-analysis. Cardiovasc Res. 2020 Jan 1;116(1):28-39. PubMed | Google Scholar
- Liu W, Cao Y, Dong L, Zhu Y, Wu Y, Lv Z et al. Periodontal therapy for primary or secondary prevention of cardiovascular disease in people with periodontitis. Cochrane Database Syst Rev. 2019 Dec 31;12(12):CD009197. PubMed | Google Scholar
- Nazir M, Al-Ansari A, Al-Khalifa K, Alhareky M, Gaffar B, Almas K. Global Prevalence of Periodontal Disease and Lack of Its Surveillance. Scientific World Journal. 2020 May 28;2020:2146160. PubMed | Google Scholar
- Nazir MA. Prevalence of periodontal disease, its association with systemic diseases and prevention. Int J Health Sci (Qassim). 2017 Apr-Jun;11(2):72-80. PubMed | Google Scholar
- Boos CJ, Lip GY. Elevated high-sensitive C-reactive protein, large arterial stiffness and atherosclerosis: a relationship between inflammation and hypertension? J Hum Hypertens. 2005 Jul;19(7):511-3. PubMed | Google Scholar
- Paraskevas S, Huizinga JD, Loos BG. A systematic review and meta-analyses on C-reactive protein in relation to periodontitis. J Clin Periodontol. 2008 Apr;35(4):277-90. PubMed | Google Scholar
- Del Pinto R, Pietropaoli D, Munoz-Aguilera E, D'Aiuto F, Czesnikiewicz-Guzik M, Monaco A et al. Periodontitis and Hypertension: Is the Association Causal? High Blood Press Cardiovasc Prev. 2020 Aug;27(4):281-289. PubMed | Google Scholar
- Chaudhry A, Kassim NK, Zainuddin SLA, Taib H, Ibrahim HA, Ahmad B et al. Potential Effects of Non-Surgical Periodontal Therapy on Periodontal Parameters, Inflammatory Markers, and Kidney Function Indicators in Chronic Kidney Disease Patients with Chronic Periodontitis. Biomedicines. 2022 Oct 29;10(11):2752. PubMed | Google Scholar
- Surma S, Romańczyk M, Witalińska-Łabuzek J, Czerniuk MR, Łabuzek K, Filipiak KJ. Periodontitis, Blood Pressure, and the Risk and Control of Arterial Hypertension: Epidemiological, Clinical, and Pathophysiological Aspects-Review of the Literature and Clinical Trials. Curr Hypertens Rep. 2021 May 7;23(5):27. PubMed | Google Scholar
- Buckley DI, Fu R, Freeman M, Rogers K, Helfand M. C-Reactive Protein as a Risk Factor for Coronary Heart Disease: A Systematic Review and Meta-analyses for the U.S. Preventive Services Task Force. Annals of internal medicine. 2009;151:7. Google Scholar
- Roca-Millan E, González-Navarro B, Sabater-Recolons MM, Marí-Roig A, Jané-Salas E, López-López J. Periodontal treatment on patients with cardiovascular disease: Systematic review and meta-analysis. Med Oral Patol Oral Cir Bucal. 2018 Nov 1;23(6):e681-e690. PubMed | Google Scholar
- Löe H. Oral hygiene in the prevention of caries and periodontal disease. Int Dent J. 2000 Jun;50(3):129-39. PubMed | Google Scholar
- Leong XF, Ng CY, Badiah B, Das S. Association between hypertension and periodontitis: possible mechanisms. ScientificWorldJournal. 2014 Jan 8;2014:768237. PubMed | Google Scholar
- Szczepaniak P, Mikołajczyk TP, Cześnikiewicz-Guzik M, Guzik TJ. Periodontitis as an inflammatory trigger in hypertension: From basic immunology to clinical implications. Kardiol Pol. 2021;79(11):1206-1214. PubMed | Google Scholar
- Campbell NRC, Paccot Burnens M, Whelton PK, Angell SY, Jaffe MG, Cohn J et al. 2021 World Health Organization guideline on pharmacological treatment of hypertension: Policy implications for the region of the Americas. Lancet Reg Health Am. 2022 May;9:100219. PubMed | Google Scholar
- Unger T, Borghi C, Charchar F, Khan NA, Poulter NR, Prabhakaran D et al. 2020 International Society of Hypertension Global Hypertension Practice Guidelines. Hypertension. 2020 Jun;75(6):1334-1357. PubMed | Google Scholar
- Bui FQ, Almeida-da-Silva CLC, Huynh B, Trinh A, Liu J, Woodward J et al. Association between periodontal pathogens and systemic disease. Biomed J. 2019 Feb;42(1):27-35. PubMed | Google Scholar