The first experience in Africa: urinary artificial sphincter type ZSI 375 PF for treatment of stress urinary incontinence in men
Ali Barki, Mohammed Aynaou, Amine El Houmaidi, Paapa Dua Boateng, Tarik Mhanna
Corresponding author: Ali Barki, Department of Urology, Mohammed VI University Medical Center, Mohammed First University, Oujda, Morocco 
Received: 24 Dec 2019 - Accepted: 04 Feb 2020 - Published: 06 Feb 2020
Domain: Urology
Keywords: Urinary incontinence, artificial sphincter, ZSI 375
©Ali Barki et al. PAMJ Clinical Medicine (ISSN: 2707-2797). This is an Open Access article distributed under the terms of the Creative Commons Attribution International 4.0 License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Cite this article: Ali Barki et al. The first experience in Africa: urinary artificial sphincter type ZSI 375 PF for treatment of stress urinary incontinence in men. PAMJ Clinical Medicine. 2020;2:36. [doi: 10.11604/pamj-cm.2020.2.36.21389]
Available online at: https://www.clinical-medicine.panafrican-med-journal.com//content/article/2/36/full
Case report 
The first experience in Africa: urinary artificial sphincter type ZSI 375 PF for treatment of stress urinary incontinence in men
The first experience in Africa: urinary artificial sphincter type ZSI 375 PF for treatment of stress urinary incontinence in men
Ali Barki1,&, Mohammed Aynaou1, Amine El Houmaidi1, Paapa Dua Boateng1, Tarik Mhanna1
1Department of Urology, Mohammed VI University Medical Center, Mohammed First University, Oujda, Morocco
&Corresponding author
Ali Barki, Department of Urology, Mohammed VI University Medical Center, Mohammed First University, Oujda, Morocco
Urinary incontinence is a disastrous complication in men following definitive treatement of prostate cancer or less commonly prostate surgery for benign conditions. In cases where conserative management fails, surgical intervention is indicated. We report our experience in Morocco with artificial sphincter type ZEPHYR ZSI 375 PF in patient with urinary incontinence after radical prostactemy (RP), with discussion of the factors that affect the response rate.
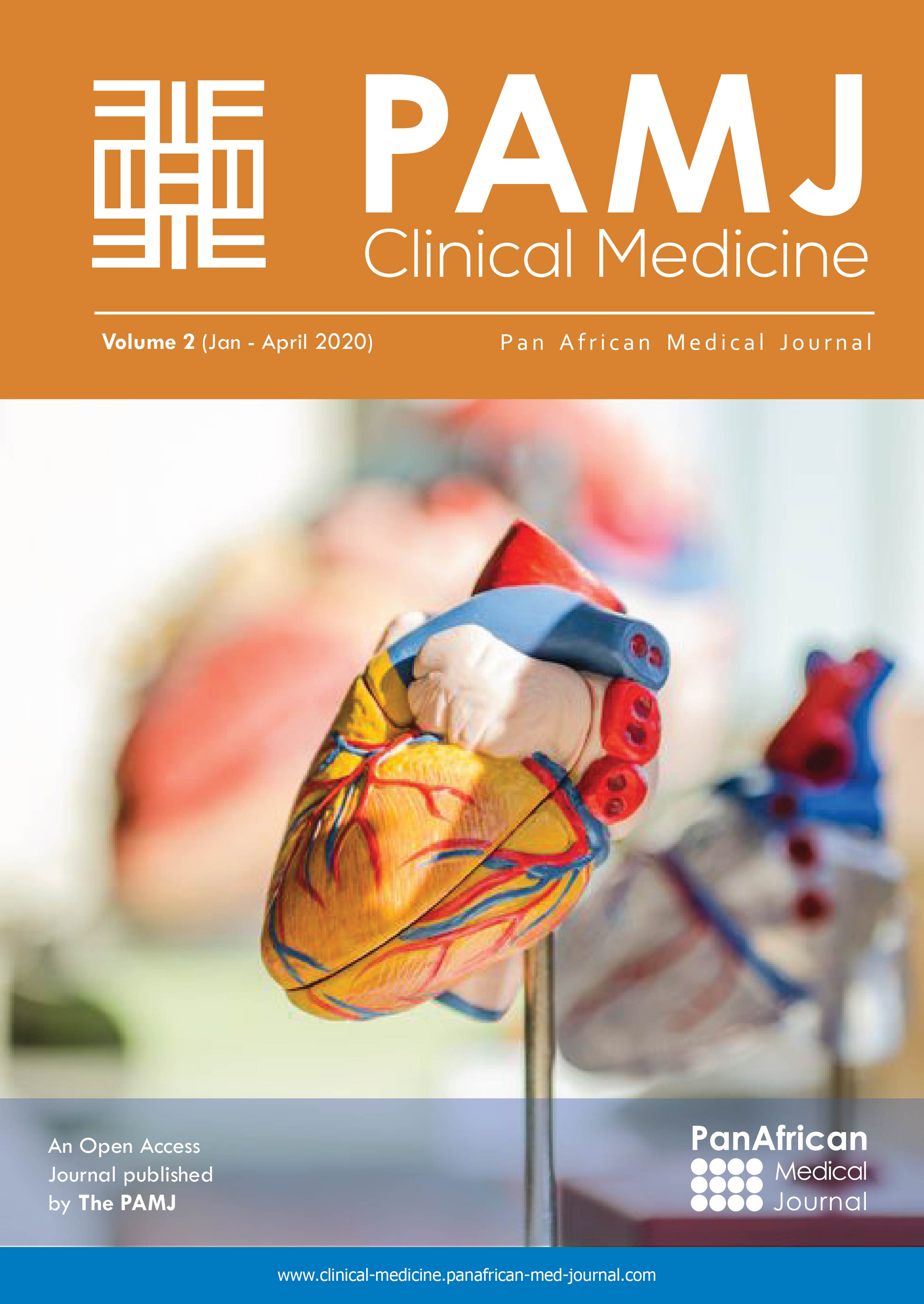
The artificial urinary sphincter (AUS) was first introduced in 1973 for the treatment of male stress urinary incontinence and became the treatment of choice for male urinary incontinence [1-3]. AUS insertion remains a complex procedure with a risk of complications such as erosion, infection and mechanical failure. Throughout the years, there have been improvements in AUS quality, with the idea of double cuffs and new surgical approaches with improved outcomes and decreased complications [3-8]. The quality of the sphincter's preparation, the connection of components and surgeon's experience during the procedure are the key factors for success as the learning curve is long. Most of the sphincter insertions are carried out by occasional operators (1 to 3 procedures per year). Less than 10% of AUS insertions are performed by surgeons with experience of at least 100 procedures in the USA [9]. The risk of revision surgery increases from 13 to 24% [10]. ZSI 375 (Zephyr Surgical Implants, Geneva, Switzerland) was designed to facilitate AUS insertion (Figure 1). The cuff is adjustable around the urethra. It´s pre-filled. The pressure can be increased to improve the patient's continence. It has no abdominal reservoir to reduce the operating time and to avoid abdominal incision and dissection in scarred retroperitoneum [11-13]. We report today the first experience in Africa, with the installation of a pre-filled artificial sphincter type ZCI 375 (PF) in a patient followed for urinary incontinence after radical prostatectomy.
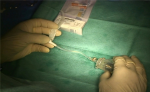
Patient: Mr. C.A, aged 75, operated for a radical prostatectomy in 2013, followed without local or regional recurrence. The patient has benefited from 15 sessions of reeducation but he still has urinary incontinence after RP. He wears 5 pads daily. ECBU is sterile. A urodynamic examination objectified a common complication, the bladder with a capacity of 260ML with presence of urinary leakage at 150ML filling, exclude an overactive bladder and a profilometry is low at 22cmHg (Figure 2). However, cystoscopy was normal without urethral or bladder abnormalities. A pose of an artificial sphincter typer zephyr is indicated. He is hospitalized 5 days after his operation. Ablation of the bladder catheter is done in D-2 of postoperative, whereas the sphincter was activated only 8 weeks later. At 3 months of follow-up, the patient is cleanly improved, absence of urinary leaks and no signs of inflammation or infection.
The device: ZCI 375 PF is AUS mainly made from medical grade silicone. It works like a typical artificial sphincter. It´s made up of a cuff connected by kink-resistant tubing to a pump unit. The inflatable and adjustable cuff molded curved fits around the urethra. The pump with the pressure-regulating tank is placed in the scrotum. After activation of the AUS, the issued pressure in the hydraulic circuit can be increased or decreased to improve patient continence [14].
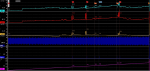
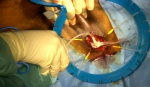
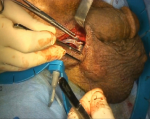
Surgical technique: the implantation procedure was carried out under general anesthesia with patient in the lithotomy position. A 16F Foley catheter was placed for guidance and to calibrate the urethra. The surgical technic consists of a perineal incision for cuff placement (Figure 3, Figure 4). A 12CH Foley catheter was inserted at the end of the procedure for 24 hours. Patients were discharged 24-48 hours after the procedure. The device was activated 8 weeks later [14].
Assessment of postoperative continence: urine analysis, bladder ultrasonography to evaluate residual urine volume and flow rate measurements must be done after sphincter activation in the following chronological order: 3 months, 6 months after activation and then every year. Success is defined as social continence (zero to one pad per day) and improvement as a decrease in daily pads use (usually two pads per day) [14].
AMS 800 AUS is currently considered the gold standard treatment for male severe urinary incontinence (SUI), but it has several drawbacks such as complexity of preparation and the procedure. Last version from 1983 improved the design of the cuff to reduce the potential for creasing and fracture leading to leakage and there is no possibility to readjust the cuff in case of urethral atrophy [15]. In case of poor result on continence, issued pressure can't be adjusted, the AMS pressure-regulating balloon has to be changed and the patient has to undergo a new procedure. ZSI 375 has been developed to overcome the lacks of AMS 800. A recent study from Germany, Poland, Italy, Spain, England and Colombia reports high success rates of AMS 800. All implanters reported the implantation procedure being very simple and straightforward and the success rate confirms that a short learning is required to achieve mastery. The results are in line with those of Staerman et al. ZCI 375 previous series outcomes have shown excellent short-term results, which ranged from 87 to 94.2% of success. The reported meantime of complication onset for AUS is 19.8 months for erosion, 29.6 months for atrophy and 68.9 months for mechanical complications [16]. Erosion risk and rate must be discussed and accepted by the patient. Radiotherapy is a very well-known adverse factor for insertion of the sphincter. In other publications, urethral erosion ranges from 2% to 17% of patients [17,18]. Suspicion of one spontaneous reactivation occurred at the same time AMS withdrew its sphincter AMS 800 from the worldwide market because of spontaneous activation in many pumps. Zephyr Surgical Implants have added a security system, which locks the sphincter in a safer deactivated position to avoid this kind of complication. The armed tubing was reinforced in 2011 to avoid kinking, thus making the tubing resistant to breakage. Usually, surgeons are not fond of preparing the AUS, filling the components with a saline solution and removing air bubbles. One sphincter required revision surgery because of a hydraulic circuit leakage of an unknown cause. The ZSI 375 now comes pre-filled, ready to be implanted and the hydraulic circuit is reinforced to reduce the risk of long-term leakage [19].
The presented case has only 3 months follow-up, but the results remain favorable, the patient has not shown any complication regarding the surgery. The quality of ZSI 375 has improved over the last five years. The reinforced hydraulic circuit of ZSI 375 PF (pre-filled) has been available since March 2015 and has reduced the operating time. The ZSI 375 device is safe, effective and the follow-up was long enough to identify all potential complications.
The authors declare no competing interests.
All the authors have read and agreed to the final manuscript.
Figure 1: the artificial urinary sphincter ZSI 375 PF (pre-filled)
Figure 2: a urodynamic tracing objectified bladder with a capacity of 260ML with presence of urinary leakage at 150ML filling
Figure 3: the implantation of the ZSI 375 PF at scrotal incision
Figure 4: the implantation of the ZSI 375 PF at inguinal incision
- Scott FB, Bradley WE, Timm GW. Treatment of urinary incontinence by implantable prosthetic sphincter. Urology. 1973;1(3):252-9. PubMed | Google Scholar
- Herschorn S, Bruschini H, Comiter C, Grise P, Hanus T, Kirschner-Hermanns R et al. Surgical treatment of stress incontinence in men. Neurourol Urodyn. 2010;29(1):179-90. PubMed | Google Scholar
- Ostrowski I, Śledź E, Ciechan J, Golabek T, Bukowczan J, Przydacz M et al. Current interventional management of male stress urinary incontinence following urological procedures. Cent European J Urol. 2015;68(3):340-347. PubMed | Google Scholar
- Montague DK. Artificial urinary sphincter: long- term results and patient satisfaction. Adv Urol. 2012;2012:835290. PubMed | Google Scholar
- Elliott DS, Barrett DM. Mayo Clinic long-term analysis of the functional durability of the AMS 800 artificial urinary sphincter: a review of 323 cases. J Urol. 1998;159(4):1206-8. PubMed | Google Scholar
- Brito CG, Mulcahy JJ, Mitchell ME, Adams MC. Use of a double cuff AMS800 urinary sphincter for severe stress incontinence. J Urol. 1993;149(2):283-5. PubMed | Google Scholar
- Guralnick ML, Miller E, Toh KL, Webster GD. Transcorporal artificial urinary sphincter cuff placement in cases requiring revision for erosion and urethral atrophy. J Urol. 2002;167(5):2075-8. PubMed | Google Scholar
- Wilson SK, Delk II Jr, Henry GD, Siegel AL. New surgical technique for sphincter urinary control system using upper transverse scrotal incision. J Urol. 2003;169(1):261-4. PubMed | Google Scholar
- Lai HH, Hsu EI, Teh BS, Butler EB, Boone TB. 13 years of experience with artificial urinary sphincter implantation at Baylor College of Medicine. J Urol. 2007;177(3):1021-5. PubMed | Google Scholar
- Wilson SK, Aliotta PJ, Salem EA, Mulcahy JJ. New enhancements of the scrotal one-incision technique for placement of artificial urinary sphincter allow proximal cuff placement. J Sex Med. 2010;7(10):3510-5. PubMed | Google Scholar
- Sandhu JS, Maschino AC, Vickers AJ. The surgical learning curve for artificial urinary sphincter procedures compared to typical surgeon experience. Eur Urol. 2011;60(6):1285-90. PubMed | Google Scholar
- Staerman F, G-Llorens C, Leon P, Leclerc Y. ZSI 375 artificial urinary sphincter for male incontinence: a preliminary study. BJU Int. 2013 Apr;111(4 Pt B):E202-6. PubMed | Google Scholar
- Lai HH, Hsu EI, The BS, Butler EB, Boone TN. 13 years of experience with artificial urinary sphincter implantation at Baylor College of Medicine. J Urol. 2007;177(3):1021-5. PubMed | Google Scholar
- Llorens C, Pottek T. Urinary artificial sphincter ZCI 375 for treatment of stress urinary incontinence in men: 5 and 7 years follow-up report. Urologia. 2017 Oct 25;84(4):263-266. PubMed | Google Scholar
- Ostrowski I, Blewniewski M, Neugart F, von Heyden B, Selvaggio O, Iori F et al. Multicentre experience with ZSI 375 artificial urinary sphincter for the treatment of stress urinary incontinence in men. Urologia. 2017;84(3):148-152. PubMed | Google Scholar
- Wilson DK, Delk JR 2nd. Ectopic placement of AMS 800 urinary control system pressure-regulating balloon. Urology. 2005;65(1):167-170. PubMed | Google Scholar
- Hajivassiliou CA. A review of the complications and results of implantation of the AMS artificial urinary sphincter. Eur Urol. 1999;35(1):36-44. PubMed | Google Scholar
- Venn SN, Greenwell TJ, Mundy AR. The long-term outcome of artificial urinary sphincters. J Urol. 2000;164(3 Pt 1):702-706. PubMed | Google Scholar
- Ostrowski I, Ciechan J, Sledz E, Dys W, Golabek T, Chłosta PL. Four-year follow-up on a Zephyr Surgical Implants 375 artificial urinary sphincter for male urinary incontinence from one urological centre in Poland. Cent European J Urol. 2018;71(3):320-325. PubMed | Google Scholar