Neurological manifestations of cytomegalovirus infection with immune reconstitution syndrome in patients with poorly-controlled HIV
Karishma Sharma, Felix Riunga, Dilraj Singh Sokhi
Corresponding author: Felix Riunga, Department of Medicine, Aga Khan University Hospital, Nairobi, Kenya 
Received: 18 Sep 2020 - Accepted: 12 Nov 2020 - Published: 09 Dec 2020
Domain: Infectious disease,Internal medicine,Neurology (general)
Keywords: Cytomegalovirus, IRIS, HIV/AIDS, neurological manifestations, polyradiculitis, stroke, ventriculitis, encephalitis
©Karishma Sharma et al. PAMJ Clinical Medicine (ISSN: 2707-2797). This is an Open Access article distributed under the terms of the Creative Commons Attribution International 4.0 License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Cite this article: Karishma Sharma et al. Neurological manifestations of cytomegalovirus infection with immune reconstitution syndrome in patients with poorly-controlled HIV. PAMJ Clinical Medicine. 2020;4:124. [doi: 10.11604/pamj-cm.2020.4.124.26139]
Available online at: https://www.clinical-medicine.panafrican-med-journal.com//content/article/4/124/full
Case series 
Neurological manifestations of cytomegalovirus infection with immune reconstitution syndrome in patients with poorly-controlled HIV
Neurological manifestations of cytomegalovirus infection with immune reconstitution syndrome in patients with poorly-controlled HIV
&Corresponding author
Cytomegalovirus reactivation is a complication of advanced HIV disease, with the main manifestation being retinitis. Other neurological manifestations are quite rare. The immune reconstitution syndrome has been reported, mainly as a uveitis. We present three cases of AIDS-associated cytomegalovirus disease with atypical neurological manifestations possibly as CMV related immune reconstitution syndrome. Three HIV positive patients, all with recent changes in their antiretroviral therapy regimen, presented with various neurological manifestations - painful polyradiculopathy, multiple ischemic infarcts and ventriculoencephalitis. Symptoms developed within 4 to 6 weeks of regimen change. CMV was diagnosed in each of the cases through cerebrospinal fluid polymerase chain reaction (CSF PCR), with supporting evidence from neuroimaging. Treatment with ganciclovir resulted in clinical improvement in all cases. New onset neurological deficits after initiation or change of ART should raise the suspicion for cytomegalovirus immune reconstitution inflammatory syndrome. Early diagnosis and prompt treatment of neurological CMV may achieve good outcomes with resolution of neurologic signs and symptoms.
Cytomegalovirus (CMV) is a ubiquitous herpes virus with an estimated global seroprevalence of greater than 80% [1]. Humans are the only reservoir, with transmission occurring through direct contact with saliva, blood, semen or vaginal secretions [2]. Viral reactivation is seen in immunocompromised patients including those with HIV/AIDS, especially with a CD4 count of less than 100 cells/mm3. The most common manifestations are retinitis and gastrointestinal disease [3]. Neurological manifestations are rare, with the most common being painful polyradiculopathy/radiculomyelitis [4]. CMV immune reconstitution inflammatory syndrome (IRIS) is even rarer and is mainly described as a uveitis [5]. Diagnosis is made by the characteristic presentation, radiological patterns and the demonstration of CMV nuclear material in the cerebrospinal fluid (CSF) or in tissue. A positive CMV polymerase chain reaction (PCR) in CSF is highly sensitive and specific for CMV neurologic disease [6].
We report three consecutive cases that had confirmed AIDS-associated CMV but presented with atypical neurological presentations due to CMV IRIS.
Patient A
A 30-year-old female presented with a five-week history of progressive painless lower limb weakness, back pain, stool and urinary incontinence, as well as visual blurring. She had been diagnosed with HIV 18 months prior at an outside facility (CD4 count and viral load not available to us). Her initial antiretroviral therapy (ART) regimen was tenofovir/emtricitabine/efavirenz, though she reported being poorly adherent. Her health care provider changed the regimen to tenofovir/lamivudine/atazanavir/ritonavir three weeks prior to symptom onset. The neurologic examination revealed flaccid paraplegia Medical Research Council (MRC) muscle power scale score of 0/5, with a T12 sensory level. She was completely blind in her left eye with decreased visual acuity in the right eye (6/12).
Work up for tuberculosis, including contrast enhanced chest and abdominal computed tomography (CT), was negative. Contrast-enhanced MRI scan of the neuraxis was remarkable for arachnoiditis to T6. Ophthalmology review confirmed bilateral CMV retinitis worse on the left. CSF was significant for lymphocytic pleocytosis with elevated protein and a positive quantitative CMV PCR. She received intravenous and intravitreal ganciclovir for 3 weeks. Repeat CSF CMV PCR testing at the end of this period was negative. She was discharged on oral valganciclovir which she took until her CD4 count was above 100 cells/mm3. Over the course of the following year, she had almost complete resolution of lower limb weakness with the most recent power grade being 4+.
Patient B
A 42-year-old female was transferred from a neighbouring country to our care with a one-week history of right-sided weakness and inability to walk. She was known to have hypertension as well as HIV. Her treatment had been changed to tenofovir/lamivudine/atazanavir/ritonavir one month prior to the symptom onset, the reason for which was not clear. Her initial CD4 count, viral load and previous ART regimen were not available to us. On neurological examination she had an upper-motor-neurone right facial nerve palsy with right hemiparesis MRC grade 4/5 in both limbs.
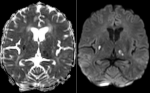
Magnetic resonance imaging (MRI) scan of the brain revealed multiple acute ischaemic infarcts of the right basal ganglia, left thalamus, pons, right medulla and left cerebellar hemisphere. Also noted were large non-restricting periventricular white matter hyperintensities in the middle cerebellar peduncle and cerebellar hemisphere (Figure 1). She was commenced on standard stroke treatment. Despite this, two days after admission she developed sudden-onset aphasia and worsening right limb weakness MRC grade 2-3/5. Repeat MRI brain scan revealed new infarcts in the left inferior frontal lobe and left lateral pons.
Additional investigations for recurrent stroke in the context of HIV were carried out (Table 1). Significantly, the CMV quantitative PCR was positive and ophthalmology review was consistent with CMV retinitis in the right retina. She was treated with intravenous ganciclovir. After 2 weeks of therapy, a repeat CMV quantitative PCR in CSF was negative. She was discharged on oral valganciclovir and stroke preventative medication and has since regained power and speech.
Patient C
A 54-year-old male presented with a one-month history of progressive cognitive decline and right-sided hemiparesis. He had a past history of hypertension. He was diagnosed with HIV two months prior to onset of these symptoms. His initial CD4 count was 16 cells/mm3 with a HIV viral load of 58000 copies/mL. His initial regimen was tenofovir/lamivudine/efavirenz but this was later changed to zidovudine/lamivudine and atazanavir/ritonavir, ostensibly due to the cognitive decline thought to be due to efavirenz. The neurological examination was significant for dysphasia, hypertonia more marked on the right, mild right pyramidal weakness and bilateral hyperreflexia including upper and lower limb clonus. Fundoscopy was normal.
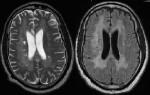
MRI brain scan showed extensive periventricular and white matter non-enhancing hyperintensities (Figure 2) without restricted diffusion, with the main differential diagnosis being progressive multifocal leukoencephalopathy. However, CSF studies were negative for John Cunningham (JC) virus PCR but positive for CMV quantitative PCR. The working diagnosis was of CMV IRIS. He was treated with intravenous ganciclovir for three weeks, then switched to oral valganciclovir. We did not give steroids as he improved with this treatment. Although his hemiparesis and dysphasia persisted, there was improvement in his cognitive status and he was transferred for ongoing rehabilitation therapy.
Radiculopathy, vascular disease/strokes, and ventriculitis/encephalitis are rare manifestations of CMV neurological disease [3]. CMV IRIS has been described and is commonly reported as a uveitis or vitritis [5] rather than involvement of other neurological structures as was the case in the patients presented. Patient A presented as a painless polyradiculopathy which is more typical for tuberculous arachnoiditis. Though quite rare overall, CMV-related polyradiculopathy is the most common manifestation of CMV in the neurological system [1,4]. It is described as a painful neuropathy and outcomes have been variable, with recovery of neurological function rather uncommon [1,3]. Her presentation was probably in keeping with CMV IRIS given the temporal association between the change of ART regimen and the onset of symptoms.
The eventual diagnosis of patient B was delayed as diagnosis of CMV-related stroke/vasculitis is particularly difficult in HIV/AIDS patients due to the wide range of differential diagnoses, including other opportunistic infections as well as traditional cardiovascular risk factors for stroke [7,8]. CMV end-organ disease has been suggested as a risk factor for ischemic stroke [9]. Similar to the first patient, there was a temporal association between change of regimen and onset of symptoms. CMV IRIS presenting as stroke is quite rare, with only one other case report in the literature [10]. In both these cases, development of symptoms was relatively rapid, within 4-6 weeks of drug initiation, which supports the diagnosis of IRIS. There were missed opportunities to undertake fundoscopy early on to detect CMV retinitis. Guidelines advocate for this assessment in cases where CD4 counts are below 100 cells/mm3 [11].
The absence of retinitis in patient C was interesting as the vast majority of those with CMV encephalitis will have concurrent retinitis as well as other end organ disease [12]. CMV encephalitis may present as a subacute cognitive decline or more acutely with delirium, cranial nerve palsies, nystagmus and ataxia [13]. Focal neurological deficits including pyramidal weakness have been described [3,12]. In addition to T2 white matter hyperintensities, restricted diffusion may also be seen on MRI imaging [14]. The presentation in this patient would also potentially fit CMV IRIS given the rapid drop in HIV viral load in association with symptom evolution. All three cases had relatively good outcomes once antiviral therapy was initiated and appropriate ART continued.
In summary, CMV disease should be a differential diagnosis in patients with neurological manifestations with a CD4 count of less than 100 cells/mm3. New onset neurological deficits after initiation or change of ART should raise the suspicion for CMV IRIS. Fundoscopy in those with CD4 counts <100 cells/mm3 should not be overlooked. Early diagnosis and prompt treatment of neurological CMV achieves good outcomes with complete resolution of neurologic signs and symptoms in some cases.
What is known about this topic
- New onset neurological deficits after initiation or change of ART should raise the suspicion for CMV IRIS;
- Fundoscopy should be performed in all patients with CD4 counts of less than 100/mm3 to detect CMV end organ involvement early in the disease.
What this study adds
- Early diagnosis and prompt treatment of neurological CMV may achieve good outcomes with resolution of neurologic signs and symptoms.
The authors declare no competing interests.
KS, FR and DSS all contributed to the formulation, drafting and revision of this case series. All the authors have read and agreed to the final manuscript.
Table 1: summary of all cases with laboratory data
Figure 1: T2/flair hyperintensities
in the left putamen and right thalamus with restricted diffusion in the diffusion
weighted images in keeping with acute infarcts
Figure 2: diffusion weighted images of the MRI depicting extensive periventricular non enhancing hyperintensities
- Crumpacker CS. Mandell, Douglas and Bennett´s principles and practice of infectious diseases. 2016. p1738-60.
- Baldwin KJ, Cummings CL. Herpes virus infections of the nervous system. Continuum (NY). 2018;24(5):1349-69. PubMed | Google Scholar
- Anders HJ, Goebel FD. Neurological manifestations of cytomegalovirus infection in the acquired immunodeficiency syndrome. Int J STD AIDS. 1999;10(3):151-61. PubMed | Google Scholar
- Anders H, Goebel F. Cytomegalovirus polyradiculopathy in patients with AIDS. 1998;345-52. Google Scholar
- Post MJ, Thurnher M, Clifford DB, Nath A, Gonzalez R, Gupta R et al. CNS-immune reconstitution inflammatory syndrome in the setting of HIV infection, part 2: discussion of neuro-immune reconstitution inflammatory syndrome with and without other pathogens. Am J Neuroradiol. 2013 Jul;34(7):1308-18. PubMed | Google Scholar
- Bestetti A, Pierotti C, Terreni M, Zappa A, Vago L, Lazzarin A et al. Comparison of three nucleic acid amplification assays of cerebrospinal fluid for diagnosis of cytomegalovirus encephalitis. J Clin Microbiol. 2001;39(3):1148-51. PubMed | Google Scholar
- Dobbs MR, Berger JR. Stroke in HIV infection and AIDS. Expert Rev Cardiovasc Ther. 2009;7(10):1263-71. PubMed | Google Scholar
- Benjamin LA, Bryer A, Emsley HCA, Khoo S, Solomon T, Connor MD et al. Review HIV infection and stroke:current perspectives and future directions. Lancet Neurol. 2012;11(10):878-90. PubMed | Google Scholar
- Kamtchum-Tatuene J, Al-Bayati Z, Mwandumba HC, Solomon T, Christmas SE, Benjamin LA. Serum concentration of anti-cytomegalovirus IgG and ischaemic stroke in patients with advanced HIV infection in Malawi. PLoS One. 2018;13(11):1-10. PubMed | Google Scholar
- Anderson AM, Fountain JA, Green SB, Bloom SA, Palmore MP. Human immunodeficiency virus - associated cytomegalovirus infection with multiple small vessel cerebral infarcts in the setting of early immune reconstitution. J Neurovirol. 2010 Mar;16(2):179-84. PubMed | Google Scholar
- Panel on Antiretroviral Guidelines for Adults and Adolescents. Guidelines for the Prevention and Treatment of Opportunistic Infections in HIV-Infected Adults. Dep Heal Hum Serv. 2018;408.
- Arribas J, Storch G, Clifford D, Tselis A. Cytomegalovirus encephalitis. Ann Intern Med. 1996;125(7):577-87. PubMed | Google Scholar
- Torok ME. Human immunodeficiency virus associated central nervous system infections. Pract Neurol. 2005;5:334-49. Google Scholar
- Cho SM, Mays M. Images in clinical neurology restricted diffusion MRI lesions in HIV-associated CMV encephalitis. The Neurohospitalist. 2018 Jan;8(1):NP3-NP4. PubMed | Google Scholar

.gif)