Oral manifestations of herbal medicine induced Steven Johnson syndrome in 3 Nigerian paediatric patients: case report
Aderonke Omolola Oluwo, Folakemi Olutoyin Irewole-Ojo, Cecilia Abimbola Mabogunje
Corresponding author: Aderonke Omolola Oluwo, Dental Department, Massey Street Children Hospital, Lagos, Nigeria 
Received: 03 Oct 2020 - Accepted: 12 Nov 2020 - Published: 12 Nov 2020
Domain: Stomatology
Keywords: Herbal medicine, oral lesions, Steven Johnson Syndrome, children
©Aderonke Omolola Oluwo et al. PAMJ Clinical Medicine (ISSN: 2707-2797). This is an Open Access article distributed under the terms of the Creative Commons Attribution International 4.0 License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Cite this article: Aderonke Omolola Oluwo et al. Oral manifestations of herbal medicine induced Steven Johnson syndrome in 3 Nigerian paediatric patients: case report. PAMJ Clinical Medicine. 2020;4:92. [doi: 10.11604/pamj-cm.2020.4.92.26367]
Available online at: https://www.clinical-medicine.panafrican-med-journal.com//content/article/4/92/full
Case report 
Oral manifestations of herbal medicine induced Steven Johnson syndrome in 3 Nigerian paediatric patients: case report
Oral manifestations of herbal medicine induced Steven Johnson syndrome in 3 Nigerian paediatric patients: case report
Aderonke Omolola Oluwo1,&, Folakemi Olutoyin Irewole-Ojo2, Cecilia Abimbola Mabogunje2
&Corresponding author
Steven-Johnson syndrome (SJS) is a rare medical emergency that is characterized by widespread skin and mucosal lesions. Oral mucosal involvement is seen in 90% of SJS and it is often widespread and confluent. In the majority of the cases, the symptoms are associated with an adverse drug reaction, usually to non-steroidal anti-inflammatory drugs, sulfonamides, anti-epileptics, or antibiotics. Recently, SJS associated with herbal medicine has been reported. This paper aims to report 3 cases of SJS with marked oral manifestations, following the use of local herbal medicine, diagnosed in a children´s hospital in Nigeria within a year. Three children aged 1.5, 9 and 10 years presented in our emergency unit with features of SJS, history of the use of local herbal medicine to cure an ailment was elicited in all cases before admission into our hospital. All the cases presented with cutaneous exfoliating exanthema and bullous eruptions with oral mucosa appearing as painful crusted erythematous lesions. One of the 3 cases, a male patient, presented clinically with septicaemia complicated with anaemia as well as nasal involvement. Discontinuation of herbal medicine in all cases resulted in a relief of symptoms. A multidisciplinary approach of management was instituted with resultant total recovery of patients. This case report addresses the fact that severe hypersensitivity reactions can occur with the use of herbal medicine. Emphasis should be on active vigilance in the use of herbal medicine.
Stevens-Johnson Syndrome (SJS) is a life-threatening inflammatory mucocutaneous disorder that presents with homogenous clinical features resulting in severe morbidity and mortality [1,2]. The lesions exhibit a rapid onset of epidermal detachment and erosion of the mucous membranes [2,3]. Oral manifestations of the disease, which is present in about 90% of cases [4], presents as painful erythematous crusts and erosions with a greyish-white membrane. In the majority of the cases, it has been widely reported that the symptoms of the disease are triggered by an adverse drug reaction, usually to non-steroidal anti-inflammatory drugs, sulfonamides, anti-epileptics, or antibiotics [5-7]. SJS can also be considered as infection driven disorders [8]. In children, Herpes simplex virus and Mycoplasma pneumoniae have been mostly identified. Herbal medicine induced SJS has been reported in the literature [6,7,9]. However, there is a paucity of literature on herbal medicine induced SJS in Nigerian children; despite a rapidly growing popularity in the use of herbal medicine. We report 3 cases of Steven-Johnson syndrome with marked oral presentation following the use of herbal medicine.
Three patients aged 1.5 years, 9 years and 10 years presented in our facility and were diagnosed with Steven Johnson syndrome. The diagnosis of SJS was made mainly based on the clinical findings, which included extensive erythema multiforme, purpuric lesions with vesicular bullous eruption, denuded or detached skin and positive Nikolsky´s sign, involving at least a mucous area such as eyes, nose, mouth, throat or genital. The 3 patients were up-to-date with all immunizations including varicella and measles, mumps, and rubella (MMR). Furthermore, they were subjected to various laboratory tests such as complete blood count, liver function tests, and the metabolic panel. The patients were immediately directed to stop the intake of the herbal mixtures.
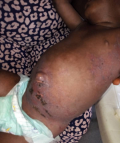
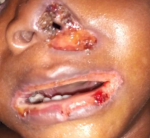
Case 1: one and a half-year-old male patient with a history of fever of 10 days duration, associated with generalized skin exfoliation of 6 days duration and skin rash all over the body. The child had been exposed to herbal medicine to treat fever. The child was previously treated at a private hospital before admission to our hospital. On examination, the boy appeared unwell with a temperature above 39.5°C, and skin detachment across the body (Figure 1). Oral mucosa appeared as reddish erosions with crusted lesions on the lower and upper lips. Multiple erythematous regions were also seen affecting buccal mucosa and tongue. There was mucocutaneous ulceration affecting the left nostrils (Figure 2). The laboratory test result is as shown below: Packed Cell Volume (PCV):16%, White Blood Cell Count (WBC): 23,700/mm3, Absolute Neutrophil Count: 10,428, Sodium: 133mmol/L, Potassium: 4.3mmol/L, Bicarbonate: 20 mmol/L, Chloride: 95 mmol/L. Renal function test (Urea: 3.1 mmol/L, Creatinine: 40mg/dL). Urine for routine and microscopic examination showed; presence of albumin, RVD: non-reactive chest X-ray: normal study.
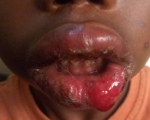
Case 2: a 10-year-old female presented with features of Steven-Johnson syndrome. On careful questioning, her parents admitted to exposing her to herbal medicine for a week to treat an ailment before she visited our facility. The herbal mixture was in 150ml airtight bottle without a label of the active ingredients. It was recommended to be effective in the treatment of fever and measles. Examination of patient revealed a generalized involvement of body including face, trunk, upper limb, lower limb including palm, soles and scalp in form of multiple wells to ill-defined discrete to confluent erythematous purpuric macules. Oral lesions appeared as extensive erythema multiforme, with the upper and lower lip and labial mucosa presenting as purpuric lesions with areas of denudation. Multiple crusted lesions with an erythematous background was observed (Figure 3). Tongue appeared depapillated with associated pain on swallowing. Laboratory test results are as shown below: PCV:32%, WBC: 12,700/mm3, lymphocytes: 36%, neutrophils:56%, Na+: 119.5mmol/L K+: 3.5mmol/L, bicarbonate: 21 mmol/L, chloride: 91.6 mmol/L, renal function test (urea: 2.9 mmol/L, creatinine: 0.4mg/dL). Liver function test (total protein: 67.7g/L and albumin 33.9g/L), RVD: non-Sreactive, chest X-ray: normal study.
Case 3: a 9-year-old male patient presented with features of Steven Johnson syndrome was referred from a private hospital to our facility. The patient was also previously exposed to herbal medicine before presentation at the emergency unit. It started as a rash which was not itchy and had a burning sensation in the oral cavity that extended into the throat. Oral examination revealed well to ill-defined, discrete, erythematous macules on the upper and lower lips, bullae and crustings were also noted. Tongue appeared depapillated and there was sloughing of the buccal mucosa. Atypical target lesions present at few places on the skin. Laboratory test results are as shown below: PCV:35%, WBC: 10,700/mm3, lymphocytes: 36%, neutrophils:62%, Na+: 139.5mmol/L, K+: 3.6mmol/L, bicarbonate: 26 mmol/L, chloride: 93.4 mmol/L, renal function test (urea: 4.0 mmol/L, creatinine: 0.7mg/dL), liver function test (total protein: 63.4g/L and albumin 36.6g/L), RVD: non-reactive, chest X-ray: normal study.
Intervention: all the 3 patients with SJS received aggressive supportive care, including intensive nursing, intravenous hydration, meticulous wound management, and appropriate specialty consultations. Prednisolone (1mg/kg/day) was administered for 3 consecutive days, IV cefuroxime 150mg/kg/day and IV metronidazole 22.5mg/kg/day for 7 days. Mouth care management was followed using guideline proposed by McPherson et al. [10] which included; daily oral review during the acute phase, white soft paraffin ointment applied to the lips every 2hours. Daily oral toileting with warm saline mouthwashes. An anti-inflammatory oral rinse or spray containing benzydamine hydrochloride was applied every 2-4 hours, particularly before eating. A potent topical corticosteroid gel; Kenalog in orabase gel triamcinolone acetonide dental paste was applied topically to affected areas four times daily. Patient 2 and 3 were placed on dexamethasone-mouthwash (10mL 0.5mg/mL; swish for 30secs and spit, four times daily for 10 days). We encouraged the continuous application of lubricant to help soften hemorrhagic lip crusts. The patients had full recovery following management and were reported as suspected cases of adverse drug reactions to a national pharmacovigilance center.
Stevens Johnson syndrome in children does not differ significantly in its etiology, clinical features and management strategies from adult SJS [11]. Oral mucosa and vermilion border involvement are seen in the majority of confirmed cases. Stomatitis and mucositis lead to impaired oral intake of food and drinks hence malnutrition, dehydration and long-term complication may lead to synechiae and esophageal stenosis. A major differential observed in SJS presenting with oral lesions is Kawasaki disease [12,13]. However, none of the 3 patients met with four or more of the clinical criteria for diagnosis of Kawasaki disease including fever for more than five days accompanied by bilateral conjunctivitis, oropharyngeal changes, cervical lymphadenopathy, polymorphous rash, and peripheral extremity changes [14]. The exact aetio-pathogenesis of Steven Johnson syndrome is not known [15]. The majority of cases are caused by an adverse reaction to a medication; however, up to 25% of cases cannot be attributed to drugs [16]. A study that reviewed the Nigerian database on SJS and toxic epidermal necrolysis (TEN) between 2004 and 2017 revealed a prevalence of 284 patients presenting with SJS and TEN. The common causative agents implicated were drugs belonging to the class of antiretroviral and antibiotics [7]. In the same study, herbal products were implicated in 5 patients [7]. The paucity of data in Nigerian children may be due to under-reporting of herbal medicines by the clinicians. SJS is classified as secondary to drugs when the patient has a history of intake of an offending drug within eight weeks before the onset of symptoms [17]. The 3 cases reported gave a positive history of intake of herbal medicine one week before symptoms.
In children presenting with SJS, an infectious causative agent can be suspected on the grounds of various clinical, laboratory, and radiological arguments [8]. Several studies have also reported infection by Mycoplasma pneumoniae to be a common trigger of SJS especially in children [8,18,19]. In our report, Mycoplasma pneumoniae was ruled out because the chest X-rays showed no abnormality. Studies have also identified viruses as a common causative agent in SJS, herpes virus has been the main virus implicated [8,16]. Routine virology study was not carried out on these patients because of the low resource setting and laboratory reports did not suggest an ongoing viral infection. The understanding of herbal mixtures involvement in the etiology of SJS has remained limited so far. This may be due to the uncertain mixture of ingredients, which makes it difficult to implicate the particular ingredient involved in the disease [6]. There have been previous reports on herbal medicine induced SJS. The authors noted recovery of the patients following the withdrawal of the herbal mixture [6,9]. All 3 patients presented in our report showed significant improvements following the withdrawal of herbal medicine. Herbal medications carry a mixture of ingredients that originate from plants and animals [9,20]. There is a rising popularity in the use of herbal medicine with claims of superior cure of ailments and has been argued to be linked to cultural and economic reasons [20]. Adverse drug reaction to herbal medicine can thus be a reaction to naturally-occurring medicinal compounds, natural toxins, or to contaminants or adulterants in these medicines [9]. The major aim of early intervention in SJS is to prevent further complication and in late phases, a reconstruction of lips damaged by chronic inflammatory reaction. Treatment involves the withdrawal of the culprit drug and supportive wound care involving fluids, nutrition, pain control and prevention, and treatment of infection [5,10,16].
Herbal medicine still plays an important role in the healthcare system in Nigeria. Because of its growing popularity, establishing a causal link between SJS and herbal medicines is crucial. Our findings suggest that clinicians should be aware of the adverse effects of herbal medicine for effective pharmaco-vigilance among patients exposed to these therapies. For these reasons, information about efficacy and safety of herbal medicines is urgently required. Further studies in larger sample size across Nigeria, comparing patient characteristics with the general population, to identify any other predisposing factors to SJS is recommended.
The authors declare no competing interests.
The three authors were involved in the management of all three cases. All authors participated in the literature search, interpretation and critique of the articles reviewed. Aderonke Omolola Oluwo prepared the initial manuscript. All authors have read and approved the final version of the manuscript.
Figure 1: showing generalized body/skin rash
Figure 2: showing crusted lips and ulcerated left nostril
Figure 3: showing swelling and redness with blister formation on the lips
- Chung WH, Hung SI, Hong HS, Hsih MS, Yang LC, Ho HC et al. Medical genetics: a marker for Stevens-Johnson syndrome. Nature. 2004 Apr 1;428(6982):486. PubMed | Google Scholar
- Mockenhaupt M. The current understanding of Stevens-Johnson syndrome and toxic epidermal necrolysis. Expert Rev Clin Immunol. 2011 Nov;7(6):803-13; quiz 814-5. PubMed | Google Scholar
- Fan ZD, Qian XQ, Yu HG. Pancytopenia as an early indicator for Stevens-Johnson syndrome complicated with hemophagocytic lymphohistiocytosis: a case report. BMC Pediatr. 2014 Feb 10;14:38. PubMed | Google Scholar
- Hazin R, Ibrahimi OA, Hazin MI, Kimyai-Asadi A. Stevens-Johnson syndrome: pathogenesis, diagnosis, and management. Ann Med. 2008;40(2):129-38. PubMed | Google Scholar
- Harr T, French LE. Toxic epidermal necrolysis and Stevens-Johnson syndrome. Orphanet J Rare Dis. 2010 Dec 16;5:39. PubMed | Google Scholar
- Lim JH, Cho SH, Lee JD, Kim HS. A case of tevens-Johnson syndrome probably induced by herbal medicine. Ann Dermatol. 2018 Aug;30(4):481-483. PubMed | Google Scholar
- Oshikoya KA, Ogunyinka IA, Ogar CK, Abiola A, Ibrahim A, Oreagba IA. Severe cutaneous adverse drug reactions manifesting as Stevens-Johnson syndrome and toxic epidermal necrolysis reported to the national pharmacovigilance center in Nigeria: a database review from 2004 to 2017. Therapeutic Advances in Drug Safety. 2020 Feb;11:2042098620905998. PubMed | Google Scholar
- Leaute-Labreze C, Lamireau T, Chawki D, Maleville J, Taieb A. Diagnosis, classification, and management of erythema multiforme and Stevens-Johnson syndrome. Arch Dis Child. 2000 Oct 1;83(4):347-52. PubMed | Google Scholar
- Lim YL, Thirumoorthy T. Serious cutaneous adverse reactions to traditional Chinese medicines. Singapore Med J. 2005 Dec;46(12):714-7. PubMed | Google Scholar
- McPherson T, Exton LS, Biswas S, Creamer D, Dziewulski P, Newell L et al. British Aassociation of dermatologists' guidelines for the management of Stevens-Johnson syndrome/toxic epidermal necrolysis in children and young people, 2018. Br J Dermatol. 2019 Jul;181(1):37-54. PubMed | Google Scholar
- Gupta LK, Martin AM, Agarwal N, D´Souza P, Das S, Kumar R, Pande S et al. Guidelines for the management of Stevens-Johnson syndrome/toxic epidermal necrolysis: an Indian perspective. Indian J Dermatol Venereol Leprol. 2016 Nov-Dec;82(6):603-625. PubMed | Google Scholar
- Demir F, Abul MH, Dilber E, Orhan F, Kalyoncu M. A case of Kawasaki disease presenting with atypical cutaneous involvement and mimicking Stevens-Johnson syndrome. North Clin Istanb. 2018 Dec 10;7(1):67-70. PubMed
- Son MB, Sundel RP. Kawasaki Disease. In: Petty RE, Laxer RM, Lindsey CB, Wedderburn LR, editors. Textbook of Pediatric Rheumatology 7 Philadelphia: Elsevier. 2016;467-483.
- Newburger JW, Takahashi M, Gerber MA, Gewitz MH, Tani LY, Burns JC et al. Committee on Rheumatic Fever, Endocarditis and Kawasaki Disease; Council on Cardiovascular Disease in the Young; American Heart Association; American Academy of Pediatrics. Diagnosis, treatment, and long-term management of Kawasaki disease: a statement for health professionals from the Committee on Rheumatic Fever, Endocarditis and Kawasaki Disease, Council on Cardiovascular Disease in the Young, American Heart Association. Circulation. 2004 Oct 26;110(17):2747-71. Google Scholar
- Swamy NN, Tasneem AF. A case series on ocular manifestations in Steven Johnson Syndrome and their varied presentation-Review of literature. J Clin Res Ophthalmol. 2020;7(1): 004-007. Google Scholar
- Paulino L, Hamblin DJ, Osondu N, Amini R. Variants of erythema multiforme: a case report and literature review. Cureus. 2018 Oct 16;10(10):e3459. PubMed | Google Scholar
- Koh MJ, Tay YK. An update on Stevens-Johnson syndrome and toxic epidermal necrolysis in children. Curr Opin Pediatr. 2009 Aug;21(4):505-10. PubMed | Google Scholar
- Sassolas B, Haddad C, Mockenhaupt M, Dunant A, Liss Y, Bork K et al. ALDEN, an algorithm for assessment of drug causality in Stevens-Johnson syndrome and toxic epidermal necrolysis: comparison with case-control analysis. Clin Pharmacol Ther. 2010 Jul;88(1):60-8. PubMed | Google Scholar
- Sah R, Neupane S, Khadka S, Poudyal S, Paneru HR, Sah R et al. A case study of Stevens-Johnson syndrome-toxic epidermal necrolysis (SJS-TEN) overlap in mycoplasma pneumoniae-associated tracheobronchitis. Case Rep Infect Dis. 2019 May 30; 2019:5471765. PubMed | Google Scholar
- Mahomoodally MF. Traditional medicines in Africa: an appraisal of ten potent african medicinal plants. Evid Based Complement Alternat Med. 2013;2013:617459. PubMed | Google Scholar