Surgical management of complicated sporadic pulmonary arteriovenous malformation
Majdi Gueldich, Walid Abid, Nesrine Kallel, Najla Bahloul, Abdessalem Hentati, Imed Frikha
Corresponding author: Walid Abid, Thoracic and Cardiovascular Surgery Department, Habib Bourguiba Hospital, Sfax University, Sfax, Tunisia 
Received: 02 Oct 2020 - Accepted: 12 Nov 2020 - Published: 15 Nov 2020
Domain: Pulmonology,Thoracic surgery
Keywords: Hemothorax, PAVM, sporadic, surgery, sardiac arrest, case report
©Majdi Gueldich et al. PAMJ Clinical Medicine (ISSN: 2707-2797). This is an Open Access article distributed under the terms of the Creative Commons Attribution International 4.0 License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Cite this article: Majdi Gueldich et al. Surgical management of complicated sporadic pulmonary arteriovenous malformation. PAMJ Clinical Medicine. 2020;4:96. [doi: 10.11604/pamj-cm.2020.4.96.26354]
Available online at: https://www.clinical-medicine.panafrican-med-journal.com//content/article/4/96/full
Surgical management of complicated sporadic pulmonary arteriovenous malformation
Majdi Gueldich1, Walid Abid1,&, Nesrine Kallel2, Najla Bahloul2, Abdessalem Hentati1, Imed Frikha1
&Corresponding author
The onset of a spontaneous hemothorax may represent a life threatening emergency. Several etiologies may be involved, mainly vascular abnormalities. Pulmonary Arteriovenous Malformation (PAVM) is a rare etiology, and it is often associated with Hereditary Hemorrhagic Telangiectasia (HHT). We report the case of a young women who was presented to the emergency department with left basal chest pain and mild dyspnea. Hemodynamic and respiratory states were maintained. Vesicular murmuring was abolished on the left side. The chest roentgenogram showed a left pleural effusion with mild deviation of the mediastinum to the opposite side. Thoracocentesis found a serohematic liquid. While the patient was undergoing exploration by computed chest tomography, she represented a cardio respiratory arrest. Resuscitation was successful after few minutes. The chest CT showed a PAVM located on the upper lobe that was very enhanced after injection of the contrast product, with a feeding artery coming from the pulmonary artery. The patient underwent an emergent left upper lobectomy thorough a posterolateral thoracotomy. The PAVM was peripheral, large with a diameter of 5 cm, and ruptured on the pleural cavity. Elective controlling the hemorrhage was very difficult, so a total hilar clamping was done with a Satinsky clamp. The postoperative course was complicated with pulmonary embolism managed by anticoagulation treatment. The definitive histological management concluded to a PAVM. Retrospectively, there was no personal or familial sign of HHT, neither any anomaly of blood hemostasis. Isolated PAVM is a rare cause of spontaneous hemothorax. Rapid worsening of an initially maintained state makes the situation more critical. Rapid control of a massive bleeding is mandatory to better explore and plan surgical resection. For stable patient, endovascular treatment may represent an effective option.
The onset of a spontaneous hemothorax may represent a life threatening emergency. Management depends on the hemodynamic and respiratory conditions, and the underlying cause. Pulmonary Arteriovenous Malformation (PAVM) is a rare etiology, and it is often associated with Hereditary Hemorrhagic Telangiectasia (HHT). We report a case of a spontaneous hemothorax secondary to isolated PAVM, complicated with a cardio respiratory arrest, and managed by urgent lobectomy.
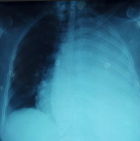
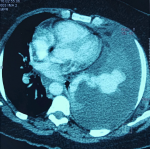
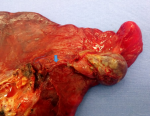
We report the case of a 43-years-old woman, with no history of comorbidity, who was presented to the emergency department with left basal chest pain and mild dyspnea. Symptomatology started three days ago. Oxygenation on ambient air was 95%, with a respiratory rate of 20 cycles per minute. The systolic blood pressure was 110 mmHg and heart rate was 95 beats per minute. Vesicular murmuring was abolished on the left side. The chest roentgenogram showed a left pleural effusion with mild deviation of the mediastinum to the opposite side (Figure 1). Thoracocentesis found a serohematic liquid. As this effusion was well tolerated by the patient, no chest drainage was performed and we decided to complete with a computed tomography CT of the chest. A few seconds after the injection of the iodinated contrast product, the patient represented a rapid worsening of the respiratory and hemodynamic condition leading to a cardio respiratory arrest. Three hypothesis was advocated to explain this cardio-pulmonary arrest: 1- an anaphylactic reaction due to the injection of an iodinated product, 2- an hypovolemic cause due to an active hemorrhage inside the pleural space, 3-an hypoxemic etiology regarding the importance of pleural effusion. Resuscitation was successful after few minutes with introduction of vasopressor drugs and mechanical ventilation. After stabilization of the vital conditions, the chest CT showed a PAVM located on the upper lobe that was much enhanced after injection of the contrast product. As showed in Figure 2, there was a feeding artery coming from the pulmonary artery. The effusion was massive with a total collapse of the hole lung. No other abnormalities were found. The patient was directly transferred to the operating room. Thorough a posterolateral left thoracotomy and after evacuation of two liters of blood, the PAVM was peripheral, large with a diameter of 5 cm, and ruptured on the pleural cavity. A left upper lobectomy was performed after total clamping of the pulmonary artery (Figure 3). The postoperative course was complicated with pulmonary embolism managed by anticoagulation treatment. The definitive histological management concluded to a PAVM. There was no personal or familial sign of HHT, neither any anomaly of blood hemostasis.
Firstly described at the end of the 19th century, Pulmonary Arteriovenous Malformation (PAVM) can be defined as a direct communication between the arterial and venous system of the pulmonary circulation, with the absence of a capillary bed [1,2]. Thus, a PAVM is composed by an afferent artery, an aneurismal part that can be a sac or a serpiginous network, and a draining vein. Depending on the configuration of the feeding vessel, the PAVM is considered as simple when there are one or more arteries originating from the same segmental pulmonary artery (80% of cases). When arteries come from several segmental arteries, PAVM is called complex [3]. As mentioned in the paper of Lacombe et al. [3], the development of a PAVM comprises several steps, starting from a ground glass nodule, passing from an abnormal communication between pre and post capillary vessels, leading to the definitive PAVM. This abnormal vascular communication may result to a significant right-to-left shunting, that can be clinically expressed by dyspnea, cyanosis, clubbing, polycythemia, and neurological central manifestation such as ischemic accidents and brain abscess [4, 5]. The natural evolution of the PAVM is often characterized by the increase of its size, exposing to the risk of rupture [3]. This complication may lead to a serious hemorrhage that can be presented by massive hemoptysis or hemothorax like in our case. This later presentation is not frequent. In a series of 143 cases of PAVM, only 5 developed an hemothorax [2, 6].
In most cases, PAVM remains asymptomatic until the fourth decade of life [4], and they are infrequently sporadic (10% in approximation). An association with Hereditary Hemorrhagic Telangiectasia (HHT) or Rendu-Osler-Weber disease is found in more than 80% of cases [2,3]. This autosomic dominant disorder is characterized by the development of multiple arterio-venous malformation in different tissue like the skin, mucosal membranes and visceral organs. When located on the lung, malformations are often multiple, bilateral and basal, contrary to sporadic cases. Taking into consideration this frequent association, HHT must be searched on the basis of Curaçao criteria (multiple muco-cutaneous telangiectases; spontaneous and recurrent epistaxis; visceral involvement; a family first degree history of HHT) [3]. In our case, no criterion is found, and PAVM is considered as sporadic. Regarding the significant morbidity and mortality, a radical treatment is mandatory when a PAVM is diagnosed, even in asymptomatic patient. Computed Tomography (CT) scan of the chest is the gold standard for the diagnostic, with the same sensitivity and specificity of the pulmonary angiography [4]. Location, size and number of the PAVM must be detailed for the choice of the appropriate treatment. Percutaneous endovascular embolisation (PEE) of the feeding artery appears to be an effective therapy in experimented hands. As endovascular approaches are in perpetual development, this elegant technique offers a good result with an average success rate of 98% [5]. Failure may be explained by the apparition of other PAVM, recanalization and development of collateral vessels [6]. PEE is possible for small PAVM in stable patient. Otherwise, a surgical treatment is mandatory. Surgery is based on lung sparing resection; so the PAVM may be removed by wedge, sublobar-resection or even lobectomy. It is indicated when the fistula is large, the patient is unstable, the PAVM occupies a big part of the lung, or in case of failure of PEE [4]. Video-thoracoscopy (VTS) is nowadays an advocated approach when possible [5]. Homma et al. reported a case of spontaneous hemothorax caused by the rupture of micro venous hemangioma, with conserved hemodynamic constants, treated by a wedge resection via VTS [7]. Lung transplantation as a treatment of multiple PAVM is reported in the literature, and is indicated for very selected cases [1,5].
Sporadic PAVM may be revealed by a spontaneous hemothorax, with significant morbidity and mortality. Management depends on hemodynamic conditions and characteristics of the PAVM. Surgical treatment should be considered as the definitive treatment. PEE is an elegant alternative in selected cases.
The authors declare no competing interests.
All the authors have read and agreed to the final manuscript.
Figure 1: the chest roentgenogram showing left pleural effusion with mild deviation of the mediastinum
Figure 2: enhanced PAVM with feeding artery originating from pulmonary artery
Figure 3: macroscopic view of the PAVM and the feeding artery (blue arrow)
- Graciela Svetliza, Alicia De la Canal, Enrique Beveraggi, Alejandro Giacoia, Claudio Ruiz, Enrique Caruso et al. B: Lung transplantation in a patient with arteriovenous malformations. J Heart Lung 2002;21(506-508). PubMed | Google Scholar
- Ference BA, Shannon TM, White RI Jr, Zawin M, Burdge CM. Life-threatening pulmonary hemorrhage with pulmonary arteriovenous malformations and hereditary hemorrhagic telangiectasia. Chest. 1994;106(5):1387-1390. PubMed | Google Scholar
- Lacombe P, Lacout A, Marcy PY, Binsse S, Sellier J, Bensalah M et al. Diagnosis and treatment of pulmonary arteriovenous malformations in hereditary hemorrhagic telangiectasia: an overview. Diagnostic and interventional Imaging. 2013;94(9):835-848. PubMed | Google Scholar
- Liechty KW, Flake AW. Pulmonary vascular malformations. Seminars in Pediatric Surgery. 2008;17(1):9-16. PubMed | Google Scholar
- Reynaud-Gaubert, Thomas P, Gaubert JY, Pietri P, Garbe L, Giudicelli R et al. Pulmonary arteriovenous malformations: lung transplantation as a therapeutic option. Eur Respir J. 1999 Dec;14(6):1425-8. PubMed | Google Scholar
- Broderick SR. Hemothorax: Etiology, diagnosis, and management. Thoracic surgery clinics. 2013;23(1):89-96. PubMed | Google Scholar
- Homma T, Yamamoto Y, Imura J, Doki Y, Yoshimura N, Senda K et al. Spontaneous hemothorax caused by pulmonary micro-venous hemangioma. The Annals of Thoracic Surgery. 2015;100(1):299-301. PubMed | Google Scholar