Immunosuppressive drugs and ganciclovir: two modalities to prevent COVID-19 severity: a case report
Meriem Ben Salem, Letaief Salima, Hamouda Mouna, Sahbi Khaled Taieb, Insaf Handous, Ben Saleh Manel, Letaief Ahmed, Aloui Sabra, Habib Skhiri
Corresponding author: Meriem Ben Salem, Department of Nephrology, Dialysis and Kidney Transplantation Fattouma Bourguiba University Hospital of Monastir, Monastir, Tunisia 
Received: 15 Mar 2021 - Accepted: 22 Apr 2021 - Published: 23 Apr 2021
Domain: Infectious disease,Nephrology
Keywords: Kidney transplant, SARS-CoV-2, immunosuppressive drugs, ganciclovir, case report
©Meriem Ben Salem et al. PAMJ Clinical Medicine (ISSN: 2707-2797). This is an Open Access article distributed under the terms of the Creative Commons Attribution International 4.0 License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Cite this article: Meriem Ben Salem et al. Immunosuppressive drugs and ganciclovir: two modalities to prevent COVID-19 severity: a case report. PAMJ Clinical Medicine. 2021;5:86. [doi: 10.11604/pamj-cm.2021.5.86.28901]
Available online at: https://www.clinical-medicine.panafrican-med-journal.com//content/article/5/86/full
Case report 
Immunosuppressive drugs and ganciclovir: two modalities to prevent COVID-19 severity: a case report
Immunosuppressive drugs and ganciclovir: two modalities to prevent COVID-19 severity: a case report
![]() Meriem Ben Salem1,&, Letaief Salima1, Hamouda Mouna1,
Meriem Ben Salem1,&, Letaief Salima1, Hamouda Mouna1, ![]() Sahbi Khaled Taieb1,
Sahbi Khaled Taieb1, ![]() Insaf Handous1, Ben Saleh Manel1, Letaief Ahmed1, Aloui Sabra1,
Insaf Handous1, Ben Saleh Manel1, Letaief Ahmed1, Aloui Sabra1, ![]() Habib Skhiri1
Habib Skhiri1
&Corresponding author
Little is known about clinical presentation, complications and therapy of SARS-CoV-2 infection in kidney transplantation patients. Herein, we report the first case of COVID-19 in kidney transplant patient in our country with excellent outcome. The patient was hospitalized for recurrent cyto megalo virus (CMV) disease treated with ganciclovir. Her immunosuppressant regimen included: mammalian target of rapamycin (mTOR) inhibitor, tacrolimus and corticosteroids. On day 35 of hospitalization, she developed a relatively mild SARS-CoV-2 infection which was detected randomly and resolved rapidly. So, it is possible that Immuno suppression can be “protective”, and some antiviral drugs such as ganciclovir may be effective against SARS-CoV-2. More data are needed to better understand SARS-CoV-2 infection in solid organ transplant patients.
A novel coronavirus strain named severe acute respiratory syndrome Coronavirus 2 (SARS-CoV-2) was described at the end of 2019 in Wuhan, China. As it spread quickly around the world. The clinical presentation of this infection can vary from an asymptomatic course to acute respiratory distress syndrome (ARDS) requiring invasive mechanical ventilation and extracorporeal membrane oxygenation, and potentially leading to death both in general population and in kidney transplant patients [1]. Little is known about presentation, risk and issues of SARS-CoV-2 infection in kidney transplantation patients, who may be at high-risk due to comorbidity and long-term immunosuppression. Since specific medications to treat this infection have not yet been revealed, researchers are working on conceiving various drugs -with at least minor action against this virus. Several antiviral drugs have been considered to be potentially successful in treating COVID-19, including lopinavir/ritonavir, oseltamivir, arbidol and ganciclovir [2]. Currently, cytokine storms are considered as the main cause of severe disease course and subsequent death due to COVID‐19, justifying why many researchers are attempting to inhibit the inflammation in lung tissues during the infection phase. Some studies proposed mTOR inhibitors as a target that could be employed for COVID-19 infection [3]. Herein, we report the first case of COVID-19 in kidney transplant patient in our country with excellent outcome despite the coinfection with SARS-CoV-2 and Cytomegalovirus (CMV).
A 36 years old woman, with a remarkable medical history of severe systemic lupus erythematosus and later chronic kidney disease secondary to lupus nephritis, underwent peritoneal dialysis for two years then switched to hemodialysis due to recurrent peritonitis. She received a renal transplant from cadaveric donor on October 02, 2019. The induction immunosuppression (IS) associated anti-thymocyte globulin, tacrolimus, mycophenolate mofetil (MMF) and steroids. As maintenance IS, she received tacrolimus 0.15mg/kg/day, MMF 0.5 g every 12 hours, and prednisone 5mg daily. This regimen´s complications included: thrombophlebitis treated by anticoagulation therapy, complicated with abdominal muscle hematoma and a large hematoma around the kidney graft requiring an urgent surgical intervention; urinary tract infection; adrenal insufficiency and recurrent CMV infections. Owing to gastrointestinal disorders and recurrent CMV infections, MMF was converted to Sirolimus 1 mg daily. Worth noting that, the hydroxychloroquine was discontinued six years ago due to toxic maculopathy: her IS regiment thus comprised of Tacrolimus 1mg twice daily, Sirolimus 1 mg per day, and Prednisone 10 mg per day. On July 23rd, 2020, she was hospitalized for a reduced visual acuity of her left eye and abdominal discomfort associated to CMV disease (CMV colitis and retinitis) treated by intravenous ganciclovir 10 mg/kg/day for 60 days. CMV viral load monitoring during antiviral therapy showed a decreasing load since the first week -but a significant lymphopenia of 0.890 x 109/L was discovered at Day 10. After five weeks of therapy, the patient displayed no signs of CMV retinitis. On day 35 of hospitalization, she presented an isolated cough. Physical examination was otherwise unremarkable (including breath sounds on chest auscultation). Her oxygen saturation was 98% when breathing ambient air.
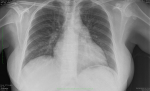
A routine blood test showed that her peripheral white cell count was 6.29 x 109/L, with a remarkable lymphopenia of 0.86 x 109/L (the previous lymphopenia initially improved with the gancilovir therapy), neutrophils count was 5.1 x 109/L, hemoglobin count was 10,15 g/dl and platelet count was 131 × 10⁹/L. She had elevated C-reactive protein (CRP) and lactate dehydrogenase (LDH) levels (33.5 mg/L and 324 U/L, respectively). Liver enzymes were slightly increased and her serum creatinine was still within the normal range. Laboratory data are reported in Table 1. Chest x-ray displayed increased lung markings with bilateral hilar congestion and a right basal ill-defined low density (Figure 1). We suspected A typical bacterial infection so we performed the serological tests of Chlamydia, mycoplasma, legionella and rickettsia. Levofloxacin 500 mg daily was administrated to the patient but it was discontinued as soon as all the serological tests came back negative (after 7 days). However, the cough disappeared 72 hours after the antibiotic therapy (Figure 2). Meanwhile, the nurse who took care of our patient was diagnosed with COVID-19 infection. As part of contact tracing, a nasopharyngeal swab was performed for our patient and it was positive for SARS-CoV-2. However, she had no complaints: her physical examination and her laboratory testing were otherwise unremarkable. Thus, we decided to keep her quarantined at her home and to give her ascorbic acid 500mg every 8 hours, Zinc 30 mg daily and vitamin D. Her immunosuppressive regiment remained unchanged. We conducted regular follow-up phone calls for two weeks to monitor her for any alarming signs of COVID-19: she has been healthy, and no symptom was reported.
We report a case of COVID-19 occurring in a kidney transplant recipient, detected randomly by the contact tracing -of a nurse who tested positive for SARS-CoV-2. The patient had a relatively mild infection with an isolated cough related to an atypical pneumonia that resolved rapidly. Cough is the classic symptom of COVID-19. It had been reported in around 50%-76% of patients (depending on the series) [4]. We retrospectively concluded that elevated levels of LDH and liver enzymes, were caused by the COVID-19 infection. Although CMV infection and ganciclovir therapy could induce these biological abnormalities, our patient´s CMV infection was managed (significantly decreased viral load) and these anomalies were resolved rapidly without reducing the dose of ganciclovir. Despite a lymphopenia, our patient presented a mild disease. However, it was reported that lymphopenia has been associated with COVID-19 severity [5]. So, we can hypothesize that she might have been protected by either the immunosuppressive drugs or the antiviral drug ganciclovir. Currently, the proposed pathologic process of severe SARS-CoV-2 infection is related to an immune over-reaction, resulting in an uncontrolled cytokines release [1]. Consequently, the use of drugs acting on immune homeostasis -such as IS- might calm the cytokine storm.
Our patient was under mTOR inhibitor rapamycin associated to tacrolimus and corticosteroids; which were indeed continued during the infection without any significant complications. Concerning mTOR inhibitors, -in addition to their function as immunosuppressants- they have multiple roles in innate immunity [3]. When associated with corticosteroids, they can significantly improve the outcome in intensive care unit patients with severe H1N1 influenza virus infection: this gives us additional evidence supporting clinical benefit of mTOR inhibitors for similar diseases -like SARS-CoV-2 infection. Some recently published articles also back-up the potential therapeutic effects of mTOR inhibitors against COVID‐19 [6]. We recommend to conduct a randomized, double‐blind, placebo‐controlled, multicenter clinical trial to prove the safety and adequacy of mTOR inhibitors in preventing COVID‐19 severity. In terms of calcineurin inhibitors (CNIs) management for kidney transplant patients during COVID-19 pandemic, heterogeneous approaches have been reported ranging from our attitude to keep them, to their discontinuation [7]. In an Italian cohort, the use of tacrolimus indicated a trend as a risk factor for both ARDS and mortality [8]. Concerning the use of corticosteroids in treating SARS-CoV-2 infection, some studies support its use at low-to-moderate dose. It could reduce death and shorten the length of in-hospital stay for gravely ill patients with SARS without inducing secondary infection and other complications [8]. Besides the IS therapy, the patient received ganciclovir in the context of CMV infection. We hypothesized that this drug could potentially prevent or treat SARS-CoV-2 infection. In the literature, few studies reported the use of ganciclovir in COVID-19 management. Hang et al. described a complete recovery in one patient who was treated with ganciclovir [9]. Recently, Haiping et al., by the use of a deep learning drug screening, had identified ganciclovir as a potential treatment for SARS-CoV-2 [10]. The efficacy of this drug against COVID-19 infection needs to be evaluated by additional clinical trials.
To conclude, we report the first case of COVID-19 in a kidney transplant recipient in our country. It reminds us that immuno compromised patients may present with atypical clinical presentations. The ideal therapeutic management is still unclear. It is possible that immunosuppression can be “protective”. Additionally, other drugs such as ganciclovir may have an effective antiviral activity against SARS-CoV-2. We need more data to better understand SARS-CoV-2 infection in solid organ transplant patients.
The authors declare no competing interests.
Ben Salem Meriem, Letaief Salima, Taieb Sahbi Khaled and Hamouda Mouna participated in the data collection and the study design and wrote the manuscript. Handous Insaf, Ben Saleh Manel, and Letaief Ahmed provided intellectual content and participated in the conception and interpretation. Aloui Sabra and Skhiri Habib provided intellectual content of critical importance to the work and reviewed the article. All authors read and approved the final manuscript.
Table 1: clinical laboratory results
Figure 1: patient's chest X-ray on day 35 of hospitalization (when she presented a cough)
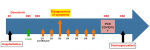
Figure 2: graphic representation of symptoms presentation, diagnostic of COVID-19, treatment, and progression of symptoms
- Banerjee D, Popoola J, Shah S, Chis Ster I, Quan V, Phanish M. COVID-19 infection in kidney transplant recipients.Kidney Int. 2020 Jun;97(6):1076-1082. PubMed | Google Scholar
- Mahmoud Y, Alireza Z, Kosar M, Zarghi A, Hosseini M, Safari S. Antiviral therapy in management of COVID-19: a systematic review on current evidence. Arch Acad Emerg Med. 2020 Apr 6;8(1):e45. PubMed | Google Scholar
- Yunfeng Z, Renfeng L, Shunai. Immunoregulation with mTOR inhibitors to prevent COVID-19 severity: a novel intervention strategy beyond vaccines and specific antiviral medicines. J Med Virol. 2020 Sep;92(9):1495-1500. PubMed | Google Scholar
- Grant MC, Geoghegan L, Arbyn M, Mohammed Z, McGuinness L, Clarke EL et al. The prevalence of symptoms in 24,410 adults infected by the novel coronavirus (SARS-CoV-2; COVID-19): a systematic review and meta-analysis of 148 studies from 9 countries. PLOS ONE. 2020;15(6): e0234765. PubMed | Google Scholar
- Tan L, Wang Q, Zhang D, Ding J, Huang Q, Tang Y et al. Lymphopenia predicts disease severity of COVID-19: a descriptive and predictive study. Signal Transduct Target Ther. 2020 Mar 27;5(1):33. PubMed | Google Scholar
- Gordon DE, Jang GM, Bouhaddou M, Xu J, Obernier K, White KM et al. A SARS-CoV-2 protein interaction map reveals targets for drug repurposing. Nature. 2020 Jul;583(7816):459-468. PubMed | Google Scholar
- Husain SA, Chang JH, Cohen DJ, Crew RJ, Dube GK, Fernandez HE et al. Early description of coronavirus 2019 disease in kidney transplant recipients in New York. J Am Soc Nephrol. 2020 Jun;31(6):1150-1156. PubMed | Google Scholar
- Bossini N, Albericin F, Delbarba E, Valerio F, Manenti C, Possenti S et al. Kidney transplant patients with SARS-CoV-2 infection: the brescia renal COVID task force experience. Am J Transplant. 2020 Nov;20(11):3019-3029. PubMed | Google Scholar
- Han X, Fan Y, Wan YL,Shi H. A diabetic patient with 2019-nCoV infection who recovered and was discharged from hospital. J Thorac Imaging . 2020 May;35(3):W94-W95. PubMed | Google Scholar
- Haiping Z, Konda MS, Yang Y, Hossain MT, Li J, Ren X et al. Deep learning based drug screening for novel Coronavirus 2019-nCov. Interdisciplinary sciences: computational Life Sciences. 2020 Sep;12(3):368-376. Google Scholar