Treatment with hydroxychloroquine vs nitazoxanide in patients with COVID-19: brief report
José Meneses Calderón, Srivatsan Padmanabhan, Fernando Manuel Carranza Salazar, David Californias Hernández, Alfredo Israel Díaz Martínez, César Humberto Botello Ortiz, Hugo Mendieta Zerón
Corresponding author: Hugo Mendieta Zerón, Faculty of Medicine, Autonomous University of the State of Mexico (UAEMéx), Toluca, Mexico 
Received: 29 Jul 2021 - Accepted: 21 Sep 2021 - Published: 23 Nov 2021
Domain: Infectious disease,Intensive care medicine,Internal medicine
Keywords: Coronavirus, hydroxychloroquine, intensive care unit, nitazoxanide
©José Meneses Calderón et al. PAMJ Clinical Medicine (ISSN: 2707-2797). This is an Open Access article distributed under the terms of the Creative Commons Attribution International 4.0 License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Cite this article: José Meneses Calderón et al. Treatment with hydroxychloroquine vs nitazoxanide in patients with COVID-19: brief report. PAMJ Clinical Medicine. 2021;7:15. [doi: 10.11604/pamj-cm.2021.7.15.30981]
Available online at: https://www.clinical-medicine.panafrican-med-journal.com//content/article/7/15/full
Treatment with hydroxychloroquine vs nitazoxanide in patients with COVID-19: brief report
José Meneses Calderón1,2, ![]() Srivatsan Padmanabhan3,4,
Srivatsan Padmanabhan3,4, ![]() Fernando Manuel Carranza Salazar5, David Californias Hernández5,
Fernando Manuel Carranza Salazar5, David Californias Hernández5, ![]() Alfredo Israel Díaz Martínez6, César Humberto Botello Ortiz6,
Alfredo Israel Díaz Martínez6, César Humberto Botello Ortiz6, ![]() Hugo Mendieta Zerón1,2,&
Hugo Mendieta Zerón1,2,&
&Corresponding author
Introduction: coronavirus pandemic has led to 1.8 million deaths worldwide as of May 1st, 2021. Nitazoxanide (NTZ) is currently being studied in several ambulatory clinical trials to control viral infections including SARS-CoV-2. This study evaluates the effect of treatment with NTZ compared to Hydroxychloroquine (HCQ) in hospitalized patients.
Methods: in this clinical study performed from May to August 2020, two groups of COVID-19 patients were compared: A) a control group treated with HCQ 200 mg PO twice a day for seven days and B) an experimental group treated with NTZ 500 mg PO every 6 hours for seven days. Student's t-test between the two groups and Relative Risk (RR) with a 95% confidence interval (CI), were calculated. In all cases a P<0.05 was considered statistically significant.
Results: twenty-seven (27) patients (females 17, males 10) treated with HCQ and 17 patients (females 8, males 9) administered NTZ were included in the study (mean age 44.6 ± 14.4 years old). At the moment of hospital admission, the following variables were statistically significant: BMI (P=0.0225) and leucocyte counts (P=0.069). In the follow-up period, the statistically significant variables were hospital length of stay (P=0.003257) and Intensive Care Units (ICU) admission with a lower RR in the group taking NTZ vs HCQ of 0.4074 (95% CI: 0.2451 to 0.5927, attributable risk (P1 - P2): 0.5926, P≤0.0001, NNT: 1.688).
Conclusion: in comparison to HCQ, NTZ significantly reduces the risk to be hospitalized in the ICU and this approach could be replicated easily in any hospital.
Coronaviruses (CoV) are positive-sense single-stranded RNA viruses in the Coronaviridae family (Coronavirinae subfamily) that infect a wide range of hosts to produce diseases ranging from the common cold to serious/fatal diseases [1]. Rapid spread of COVID-19 and the ensuing havoc has led physicians to try a variety of treatments due to the paucity of drugs available for this disease [2]. Currently approved drugs include the RDRP inhibitor, remdesivir, which is cost prohibitive for most developing countries. In addition, the WHO SOLIDARITY trial did not reveal a significant effect of remdesivir when compared to placebo in moderate to severe disease. The ACTIV trial, however, showed a small reduction in hospital length of stay (LOS) without a significant effect on mortality [3]. The results of the multi-arm SOLIDARITY trial, in addition to other clinical trials on hydroxychloroquine (HCQ) (an established therapy for several rheumatological disorders) for moderate to severe disease, did not show any effect on mortality (or mortality due to arrhythmias) [4,5].
For hospitalized patients with COVID-19 pneumonia who are mechanically ventilated, the American Thoracic Society/European Respiratory Society coordinated International Task Force suggests dexamethasone based on the RECOVERY trial data, where dexamethasone was shown to reduce mortality in patients with hypoxia [6]. Nitazoxanide (NTZ), a derivative of 5-nitrothiazole, initially used against protozoan and helminths, has also showed antibacterial and antiviral activity [7]. Because of its broad-spectrum antiviral activity [8], NTZ has been studied in several clinical trials to control viral infections [7,8,9]. In vitro activity against SARS-CoV-2 is encouraging [10] with equally encouraging initial clinical experience [9]. It is clear that more data is needed to determine its role in the management of COVID-19 in a real-life clinical setting. The NTZ dosing for anti-viral effect is higher than that used for anti-parasitic effect, and in this study we used 500 mg every 6 hours with food based on previous analysis [11-13].
The initial aim of this prospective study was to determine if there is a difference in Intensive Care Unit (ICU) utilization rates for patients with COVID-19 who were treated with HCQ in contrast to NTZ. The initial hypothesis was that the addition of NTZ in the first 48 or 72 hours of hospitalization with COVID-19 would decrease ICU admission by at least 20% based on different statistics that report the percentage of patients requiring use of intensive therapy in a threshold higher than even 30% [14].
Study design and setting: this was a prospective and descriptive study performed in the Health Institute of the State of Mexico (ISEM), Mexico, from May to August 2020. The State of Mexico has a population of 16,992,418 inhabitants, of which the ISEM serves those who do not have any type of social security, this institution has medical units in the 125 municipalities that make up the State of Mexico, of first level, second level and third level of care. The initial trial was designed to test a control group of HCQ vs HCQ+NTZ in April 2020 [15]. However, we were unable to complete recruitment for the trial due to the now retracted reports that indicated higher mortality for the HCQ treated group which resulted in an administrative halt. The small sample of NTZ only treated arm were protocol “deviations” as they did not receive HCQ in addition. The primary outcome was admission to ICU and the secondary outcomes were death or discharge alive, mechanical ventilation and LOS.
Study population: COVID-19 real-time polymerase chain reaction (qPCR) positive patients over 18 years old within the first 72 hours of diagnosis were included in this study excluding pregnant women or children, patients with prolonged corrected QT on admission electrocardiogram, patients with heart disease of any kind (heart attack, arrhythmias, hypotension with symptoms), patients who have contraindications inherent to each drug. Patients whose clinical follow-up was lost or who decided not to continue in the study were not included in the final analysis.
Sample: through a non-randomized method, due to the technical difficulty of having a person in charge in the randomization process 24 hours a day for more than one hospital, two groups were compared: A) a control group treated with HCQ 200 mg PO twice a day for seven days and B) an experimental group treated with NTZ (Daxon, Siegfried Rhein, S.A. de C.V. Mexico) 500 mg PO every 6 hours for seven days.
Data collection: patients who tested positive for COVID-19 and admitted to the hospital were evaluated for inclusion in the trial. Following signed informed consent, treatment was initiated and the taking of both drugs was supervised by the nurse staff in charge of the patients. Concomitant diseases were treated in accordance with the National and International Best Clinical Practice Guidelines. Initial vitals were recorded on arrival to the emergency unit while on room air (FiO2 21%).
Clinical information regarding the patients who received the medications were retrieved by the Research Department of the ISEM, from the respective hospitals by filling out a specific data sheet designed for this project. The severity score that was previously used for Influenza H1/N1 in Mexico was adapted for COVID-19 (Table 1).
The weight (kg) of the participants were evaluated with a minimum of clothes on a previously calibrated electronic TANITA scale. Height (m) was measured with a conventional stadiometer. The Body Mass Index (BMI) was calculated as the weight (kg) divided by the height in meters squared (m²). Blood pressure (mmHg) was measured with an electronic baumanometer. Venous blood samples were taken to measure glucose (mg/dl), uric acid (mg/dl), cholesterol (mg/dl), triacylglycerides (mg/dl) and hematic biometry. The laboratory procedures were standardized according to the International Federation of Clinical Chemistry and Medicine Laboratories (IFCC). ICU admission was registered for every patient being positive from the moment he/she crossed the door of that service, mechanical ventilation was considered as the one in which the patient was intubated, death was confirmed with the absence of clinical signs through electronic monitors.
Statistical analysis: quantitative variables were represented by measures of central tendency. First, the Kolmogorov test was performed to determine the normality of the variables and student's t-test or the Mann-Whitney U test were used as indicated through the program Social Science Statistics [16] or GraphPad Prism 9.0. Relative risk (RR), Number needed to treat (NNT) calculations for ICU admission was calculated by Fisher´s exact test using the Koopman asymptotic score to calculate the RR with 95% CI, attributable risk (Newcombe/Wilson) with GraphPad Prism version 9 for Windows, GraphPad Software, San Diego, California USA, www.graphpad.com. The COVA score to estimate the odds of hospitalization, ICU admission and death was also used [17]. In all cases, a P<0.05 was considered statistically significant. LOS was compared by unpaired t-test and by Log-Rank tests in GraphPad Prism v 9.0.
Ethical considerations: this project was authorized (code 2020-03-681) by the Research Ethics Committee of the HMPMPS, with current registration in the National Bioethics Commission (CONBIOETICA) and by the Research Committee with actual registration in the Federal Commission for Protection Against Health Risks (COFEPRIS). The research was carried out under the ethical considerations recognized by the Declaration of Helsinki (Fortaleza, Brazil, 2013), and according to the level of intervention considered a study of greater than minimal risk. In any case, an informed consent was asked to be signed prior to enrollment.
General characteristics of the study population: forty (40) patients started the HCQ arm but 10 were discarded after being prescribed with different HCQ doses from that stated in the protocol and three were excluded as they were moved to a particular Hospital. Thirty (30) patients started the NTZ arm but 11 had to be excluded because the drug was suspended for unknown reasons in a weakened and two had to be treated with lower dose after having severe gastrointestinal effects. Thus, the study analyzed 27 control patients (females 17, males 10) treated with HCQ and 17 patients (females 9, males 8) treated with NTZ. All the patients were of low-socioeconomic status. The recruitment process was stopped when there was public concern regarding the HCQ safety in COVID-19 prognosis. The mean age of the patient population was 44.6 ± 14.4 years with their general characteristics depicted in Table 2 and the main symptoms shown in Table 3. The number of patients with lymphopenia, defined as a lymphocyte count of ≤ 1500X10³ cells/mm³ were 24 (88.8%) in the HCQ group and 10 (62.5%) in the NTZ group. Patients with thrombocytopenia, defined as a platelet count lower than 150X10 cells/mm³). Were 2 (7.4%) in the HCQ group and 2 (11.7%) in the NTZ group. On initial hospital admission, the only differences on Student´s t-Test were noted in BMI (P = 0.0225) and in leucocyte counts (P = 0.069), both being higher in the HCQ group.
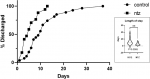
ICU admission: sixteen (59.3%) patients of the control group had to be admitted to the ICU. By contrast, in the NTZ group none of the patients needed ICU admission, but there was one (5.6%) death on day 3 of hospitalization. The RR for ICU admission in NTZ compared with the HCQ as control group was of 0.4074 (95% CI: 0.2451 to 0.5927, attributable risk (P1 - P2): 0.5926, P≤0.0001, NNT: 1.688). The COVA score for the NTZ group was 58.56, with an estimated probability (P) for hospitalization of 40.98, ICU admission/mechanical ventilation of 18.38 and Death of 3.34. The estimated P for the NTZ group as a whole for ICU admission was 18% vs 0 (Actual) and the estimated P (death) was 3.4% vs actual 5% (1 death) in the NTZ group. Finally, the difference in the LOS is shown in Figure 1. Due to the low number of patients per group it was not possible to perform stratified analyzes to adjust for the possible role of confounding variables.
Mechanical ventilation: invasive mechanical ventilation was begun in 4 (14.81%) patients from the HCQ group with a mean duration of 8.8 ± 7.5 days (range 4-20) and there were 5 deaths. By contrast, in the NTZ group no patient needed mechanical ventilation although one patient died on day 3 of admission. The RR for composite outcome of mechanical ventilation and death using HCQ vs NTZ was of 1.22 (95% CI: 0.9203 to 1.564, attributable risk (P1 - P2): 0.1697, P = 0.2414, NNT: 5.8) but was not statistically significant.
Pharmacological use: antibiotic use in the HCQ group was as follows: levofloxacin: 11 (40.74%), ceftriaxone: 9 (33.33%), cefotaxime: 5 (18.51%), meropenem: 3 (11.11%) and clarithromycin: 1 (3.7%). In the NTZ group, antibiotics used were azithromycin: 10 (55.55%), ceftriaxone: 9 (50%), levofloxacin: 5 (17.24%) and clarithromycin: 5 (17.24%). Steroid use in the HCQ group was as follows: methylprednisolone: 4 (14.81%), prednisone: 1 (3.7%) and hydrocortisone: 1 (3.7%). Among the 5 patients that died, only 2 patients had received steroids (methylprednisolone and hydrocortisone each). In the NTZ group, 5 (27.77%) patients received dexamethasone. Among other drugs, enoxaparin was prescribed in 19 (70.37%) patients in the HCQ group and in 9 (50%) patients in the NTZ group.
Length of stay (LOS): on follow-up, LOS was significantly reduced in the NTZ group with control (HCQ) mean of 11.59, NTZ mean of 5.588 and a mean reduction of -6.004 ± 2.097 compared with control, P-value of 0.0065 by two-tailed t-Test. Given the small sample size and that none of the patients in the NTZ group needed ICU admission, a subgroup analysis of LOS was also compared between control non-ICU and NTZ (non-ICU) group. Mean LOS between the two non-ICU sub-groups was still statistically significant with 8.6 for control (non-ICU) vs 5.6 for NTZ, difference of -3.048 ± 1.481 with a P-value of 0.0497, in the two-tailed t-test.
The initial clinical trial was designed as a comparison of HCQ (which was used extensively off-label when the study was first designed) vs dual therapy of HCQ and NTZ. However, due to the now retracted papers that reported higher mortality in HCQ group, the study was stopped prematurely. Serendipitously, several patients who were still treated with NTZ alone (which were protocol deviations of the original study) still showed a significant clinical effect which was further analyzed in this manuscript. NTZ treatment in hospitalized patients show a significant clinical improvement in this cohort with reduced LOS, reduced ICU admission and ventilatory requirements.
Factoring in other drugs prescribed in our patients, regarding to the administration of antibiotics, its prescription was not standardized rather was based on the known antimicrobial susceptibility of each participating service, in this case, azithromycin use was higher in the NTZ group which however has not shown any significant clinical effect in COVID-19 patients [18]. On the other hand, multiple studies including that by Albani et al. show that treatment with enoxaparin during hospital stay is associated with a lower death rate [19] and while results from larger randomized clinical trials are still pending, the patients in the HCQ group in this study received anticoagulation treatment at a higher percentage than the NTZ group.
It is striking that, since the beginning of the pandemic, NTZ was proposed as a valuable alternative against SARS-CoV-2 [20], a notion that has been reinforced based on its in vitro results [21], minimum achievable inhibitory concentration with little or no toxicity, decades of use worldwide and the low cost [22]. A previous randomized, double-blind pilot clinical trial comparing NTZ 600 mg BID versus Placebo for seven days among 50 individuals (25 each arm) with SARS-COV-2 RT-PCR+ (PCR) that were hospitalized with mild respiratory insufficiency reported two deaths in the NTZ arm and six in the placebo arm (p = 0.564). NTZ was superior to placebo when considering SSD (p < 0001), the mean time for hospital discharge (6.6 vs. 14 days, p = 0.021), and negative PCR at day 21 (p = 0.035), whereas the placebo group presented more adverse events (p = 0.04) [23]. Another analysis concluded that early COVID-19 therapy with azithromycin plus NTZ, ivermectin or HCQ in outpatient settings significantly improved COVID-19 outcomes compared to known outcomes in untreated patients [24]. Based on our data, NTZ can be envisioned as a promising alternative that urgently needs to be tested in a larger study with a recommended dose of 500 mg in tablets every 6 hours given with food.
Importantly, a recent study in a multicenter, randomized, double-blind, placebo-controlled trial with adult patients who presented up to 3 days after the onset of Covid-19 symptoms and confirmation of SARS-CoV2 infection by qPCR comparing either NTZ (500 mg suspension) or placebo, three times a day (TID), for 5 days found that early NTZ therapy was safe and reduced viral load significantly [25]. Recent finding, from a double-blind randomized multicenter study in 36 centers in the U.S. and Puerto Rico showed that NTZ treatment provided an 85% reduction in the progression to severe COVID-19 vs placebo [26]. Following this line of analysis, another study confirms a possible reduction in the intensity of COVID-19 outbreaks in health staff when NTZ is prescribed early in the clinical course of the viral infection [27].
Our study adds to the knowledge that NTZ could improve clinical outcomes in hospitalized patients with COVID-19. Given the dramatic reduction in disease severity in the hospitalized population with the dosing regimen of 500 mg tablets given every 6 hours with food, this needs to be tested in a larger randomized clinical trial in hospitalized and high-risk outpatients.
There are several important limitations to this study, the sample size was small, open label and not randomized. Given the small sample size, there were a few statistically significant differences in the population as noted below and only outcomes with the most dramatic differences met statistical significance. Despite the limitations noted above, the reduced LOS, reduced ICU admission and possibly mortality in the NTZ group point to therapeutic efficacy. Additional analysis of the NTZ group show that despite the baseline higher risk for ICU admission (18%) based on risk score (COVA), none of the treated patients ended up requiring ICU admission. Furthermore, the subgroup analysis of non-ICU admitted patients, between control and NTZ, still showed a significant overall LOS reduction. Survival analysis did not show a statistically significant difference between the groups for death, likely due to the small sample size. The generalizability of the results can be compromised for this same last condition.
What is known about this topic
- Without consensus specific treatment, COVID-19 patient care remains heterogeneous;
- Nitazoxanide has proven useful against SARS-CoV-2, being a drug with a broad safety profile and low cost.
What this study adds
- This study demonstrates the possible usefulness of nitazoxanide in reducing the risk of entering intensive care units in cases of COVID-19;
- Nitazoxanide can be tested against other alternatives in any hospital with few patients as in this study using simple statistical tests helping the medical staff to fine-tune the best scheme.
The authors declare no competing interests.
Conception and study design: José Meneses Calderón and Hugo Mendieta Zerón. Data collection: Alfredo Israel Díaz Martínez, César Humberto Botello Ortiz, Fernando Manuel Carranza Salazar and David Californias Hernández. Data analysis and interpretation: Srivatsan Padmanabhan and Hugo Mendieta Zerón. Manuscript drafting: José Meneses Calderón, Hugo Mendieta Zerón and Srivatsan Padmanabhan. Manuscript revision: José Meneses Calderón, Hugo Mendieta Zerón and Srivatsan Padmanabhan. The guarantor of the study was Hugo Mendieta Zerón. All authors approved the final version of the manuscript.
Authors thank all the health staff involved in this study who have treated the patients while putting their own lives at risk. Siegfried Rhein, S.A. de C.V. Mexico provided the medicine Daxon (Nitazoxanide). We would also like to thank Haoqi Sun and Brandon Westover for sharing the COVA score calculation details in an easy to use excel file. We also thank Kavitha Padmanabhan for edits to the final version of the manuscript.
Table 1: COVID-19 severity scale
Table 2: general characteristics of the two groups
Table 3: main symptomatology
Figure 1: length of stay survival curve (Log rank (Matel-Cox), P = 0.001); violin plot of length of stay (unpaired t-test, P = 0.0065)
- Dhama K, Sharun K, Tiwari R, Maryam D, Yashpal SM, Karam PS et al. COVID-19, an emerging coronavirus infection: advances and prospects in designing and developing vaccines, immunotherapeutics, and therapeutics. Hum Vaccines Immunother. 2020 Jun 2;16(6):1232-1238. PubMed | Google Scholar
- Vijayvargiya P, Esquer Garrigos Z, Castillo Almeida NE, Gurram PR, Stevens RW, Razonable RR. Treatment considerations for COVID-19: a critical review of the evidence (or Lack Thereof). Mayo Clin Proc. 2020 Jul;95(7):1454-1466. doi: 10.1016/j.mayocp.2020.04.027. PubMed | Google Scholar
- Beigel JH, Tomashek KM, Dodd LE, Mehta AK, Zingman BS, Kalil AC et al. Remdesivir for the treatment of Covid-19 - Final Report. N Engl J Med. 2020 Nov 5;383(19):1813-1826. doi: 10.1056/NEJMoa2007764. PubMed
- Annie FH, Sirbu C, Frazier KR, Broce M, Lucas BD et al. Hydroxychloroquine in hospitalized COVID-19 patients: real world experience assessing mortality. Pharmacotherapy. 2020 Nov;40(11):1072-1081. doi: 10.1002/phar.2467. PubMed | Google Scholar
- RECOVERY Collaborative Group, Horby P, Mafham M, Linsell L et al. Effect of Hydroxychloroquine in Hospitalized patients with Covid-19. N Engl J Med. 2020 Nov 19;383(21):2030-2040. doi: 10.1056/NEJMoa2022926. PubMed | Google Scholar
- Bai C, Chotirmall SH, Rello J, Alba GA, Ginns LC, Krishnan JA et al. Updated guidance on the management of COVID-19: from an American Thoracic Society/European Respiratory Society coordinated International Task Force (29 July 2020). Eur Respir Rev. 2020 Oct 5;29(157):200287. doi: 10.1183/16000617.0287-2020. PubMed | Google Scholar
- Shakya A, Bhat HR, Ghosh SK. Update on Nitazoxanide: a multifunctional chemotherapeutic agent. Curr Drug Discov Technol. 2018;15(3):201-213. doi: 10.2174/1570163814666170727130003. PubMed | Google Scholar
- Rossignol JF. Nitazoxanide, a new drug candidate for the treatment of Middle East respiratory syndrome coronavirus. J Infect Public Health. May-Jun 2016;9(3):227-30. doi: 10.1016/j.jiph.2016.04.001. PubMed | Google Scholar
- Meneses Calderón J, Figueroa Flores MDR, Paniagua Coria L et al. Nitazoxanide against COVID-19 in three explorative scenarios. J Infect Dev Ctries. 2020 Sep 30;14(9):982-986. doi: 10.3855/jidc.13274. PubMed | Google Scholar
- Son J, Huang S, Zeng Q, Bricker TL, Case JB et al. JIB-04 has broad-spectrum antiviral activity and inhibits SARS-CoV-2 replication and coronavirus pathogenesis. bioRxiv. 2020 Sep 25 [revised 2021 Jun 4]. doi: 10.1101/2020.09.24.312165. Google Scholar
- Padmanabhan S, Padmanabhan K. The devil is in the dosing" - targeting the interferon pathway by repositioning Nitazoxanide against COVID-19. 2020. doi:10.13140/RG.2.2.22854.83527. Google Scholar
- Lokhande AS, Devarajan PV. A review on possible mechanistic insights of Nitazoxanide for repurposing in COVID-19. Eur J Pharmacol. 2021 Jan 15;891:173748. doi: 10.1016/j.ejphar.2020.173748. PubMed | Google Scholar
- Stachulski AV, Taujanskas J, Pate SL, Rajoli RKR, Aljayyoussi G, Pennington SH et al. Therapeutic potential of nitazoxanide: an appropriate choice for repurposing versus SARS-CoV-2? ACS Infect Dis. 2021 Jun 11;7(6):1317-1331. doi: 10.1021/acsinfecdis.0c00478. PubMed | Google Scholar
- Abate SM, Ahmed Ali S, Mantfardo B, Basu B. Rate of intensive care unit admission and outcomes among patients with coronavirus: a systematic review and Meta-analysis. PLoS One. 2020 Jul 10;15(7):e0235653. doi: 10.1371/journal.pone.0235653. PubMed | Google Scholar
- Meneses Calderón J, Mendieta Zerón H, Padmanabhan, S. Treatment with Hydroxychloroquine vs Hydroxychloroquine + Nitazoxanide in COVID-19 patients with risk factors for poor prognosis: a structured summary of a study protocol for a randomised controlled trial. Trials. 2020;21:504. doi: 10.1186/s13063-020-04448-2. Google Scholar
- Free SPSS Tutorials. Social Science Statistics . 2017. Accessed on 06 February 2021.
- Sun H, Jain A, Leone MJ, Alabsi HS, Brenner LN, Ye E et al. CoVA: an acuity score for outpatient screening that predicts coronavirus disease 2019 Prognosis. J Infect Dis. 22021 Jan 4;223(1):38-46. doi: 10.1093/infdis/jiaa663. PubMed | Google Scholar
- PRINCIPLE Trial Collaborative Group. Azithromycin for community treatment of suspected COVID-19 in people at increased risk of an adverse clinical course in the UK (PRINCIPLE): a randomised, controlled, open-label, adaptive platform trial. Lancet. 2021; 397(10279):1063-1074. doi.org/10.1016/S0140-6736(21)00461-X. Google Scholar
- Albani F, Sepe L, Fusina F, Prezioso C, Baronio M, Caminiti F et al. Thromboprophylaxis with enoxaparin is associated with a lower death rate in patients hospitalized with SARS-CoV-2 infection. A cohort study. EClinical Medicine. 2020;27:100562. doi: 10.1016/j.eclinm.2020.100562. PubMed | Google Scholar
- Şimşek Yavuz S, Ünal S. Antiviral treatment of COVID-19. Turk J Med Sci. 2020 Apr 21;50(SI-1):611-619. doi: 10.3906/sag-2004-145. PubMed | Google Scholar
- Anastasiou IA, Eleftheriadou I, Tentolouris A, Tsilingiris D, Tentolouris N. In Vitro Data of Current Therapies for SARS-CoV-2. Curr Med Chem. 2020;27(27):4542-4548. PubMed | Google Scholar
- Pepperrell T, Pilkington V, Owen A, Wang J, Hill AM. Review of safety and minimum pricing of nitazoxanide for potential treatment of COVID-19. J Virus Erad. 2020 Apr 30;6(2):52-60. doi: 10.1016/S2055-6640(20)30017-0. PubMed | Google Scholar
- Blum VF, Cimerman S, Hunter JR, Tierno P, Lacerda A, Soeiro A et al. Nitazoxanide superiority to placebo to treat moderate COVID-19 - a pilot prove of concept randomized double-blind clinical trial. E Clinical Medicine. 2021 Jul;37:100981. doi: 10.1016/j.eclinm.2021.100981. PubMed | Google Scholar
- Cadegiani FA, Goren A, Wambier CG, McCoy J. Early COVID-19 therapy with azithromycin plus nitazoxanide, ivermectin or hydroxychloroquine in outpatient settings significantly improved COVID-19 outcomes compared to known outcomes in untreated patients. New Microbes New Infect. 2021 Sep;43:100915. doi: 10.1016/j.nmni.2021.100915. PubMed | Google Scholar
- Rocco PRM, Silva PL, Cruz FF, Junior MACM, Tierno PFGMM, Moura M de A et al. Early use of nitazoxanide in mild Covid-19 disease: randomized, placebo-controlled trial. medRxiv. Eur Respir J. 2021 Jul 8;58(1):2003725. doi: 10.1183/13993003.03725-2020. PubMed | Google Scholar
- Rossignol J-F, Bardin MC, Oaks JB, Bostick BG, Vora KN, Fulgencio J et al. Early treatment with nitazoxanide prevents worsening of mild and moderate COVID-19 and subsequent hospitalization. medRxiv. 2021 Jan 1;2021.04.19.21255441. doi: 10.1101/2021.04.19.21255441. Google Scholar
- Mendieta Zerón H, Meneses Calderón J, Paniagua Coria L et al. Nitazoxanide as prophylactic use to reduce the intensity of COVID-19 outbreaks in health personnel. World Acad Sci J. 2021;3:1-8. doi: 10.3892/wasj.2021.94.