Ewing's sarcoma of the bladder: a case report and review of the literature
Mounir Jamali, Youness Boukhlifi, Omar Jendouzi, Mohammed Alami, Ahmed Ameur, Rachid Tanz, Mohammed Ichou
Corresponding author: Mounir Jamali, Urology Department of the Mohammed V Military Instructional Hospital, Rabat, Morocco 
Received: 27 Oct 2021 - Accepted: 30 May 2022 - Published: 31 May 2022
Domain: Urology
Keywords: Ewing's sarcoma, bladder cancer, rare localization, case report
©Mounir Jamali et al. PAMJ Clinical Medicine (ISSN: 2707-2797). This is an Open Access article distributed under the terms of the Creative Commons Attribution International 4.0 License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Cite this article: Mounir Jamali et al. Ewing's sarcoma of the bladder: a case report and review of the literature. PAMJ Clinical Medicine. 2022;9:4. [doi: 10.11604/pamj-cm.2022.9.4.32193]
Available online at: https://www.clinical-medicine.panafrican-med-journal.com//content/article/9/4/full
Ewing's sarcoma of the bladder: a case report and review of the literature
![]() Mounir Jamali1,&, Youness Boukhlifi1,
Mounir Jamali1,&, Youness Boukhlifi1, ![]() Omar Jendouzi1, Mohammed Alami1, Ahmed Ameur1, Rachid Tanz2, Mohammed Ichou2
Omar Jendouzi1, Mohammed Alami1, Ahmed Ameur1, Rachid Tanz2, Mohammed Ichou2
&Corresponding author
Ewing's sarcoma is a very aggressive malignant bone tumor. The bladder localization of this undifferentiated form of peripheral primitive neuroectodermal tumors is extremely rare. Our patient presented with macroscopic hematuria with dysuria and suprapubic pain. After ultrasound and bladder resection, a primary bladder Ewing's sarcoma was diagnosed. The extension work-up was negative. He underwent several resections and chemotherapy with very good results. In this article, we will try to establish a comparative study of the clinical, diagnostic and therapeutic aspects of the different reported cases in order to highlight some guidelines for the management of this pathology.
Ewing's sarcoma is a malignant bone tumor first described by Dr James Ewing in 1921 and found mainly in children and young adults. It corresponds to the undifferentiated form of primary peripheral neuroectodermal tumors. Involvement of soft tissue remains extremely rare. Ewing's sarcoma of the soft tissues (EWS) was only described in 1969 by Tefft et al. [1]. The precise identification by the pathologist of this tumor among the heterogeneous group of malignant small round cell neoplasms is essential to allow optimal therapeutic management. Our objective in this study is to specify the anatomoclinical characteristics of Ewing's sarcoma and to discuss the diagnostic and therapeutic approach as well as the prognosis.
Patient information: this is Mr AB a 16-year-old with no notable history, who presented with macroscopic hematuria with suprapubic pain and dysuria in April 2020.
Clinical findings: admitted to the emergency room, where the clinical examination revealed an altered general condition with mucocutaneous pallor and discoloration of the conjunctiva.
Timeline: the patient underwent a series of biological and radiological paraclinical examinations which allowed the diagnosis of his disease before starting his management with surgery and chemotherapy.
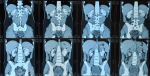
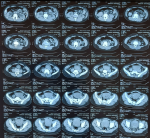
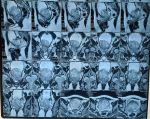
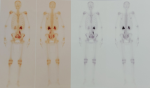
Diagnostic assessment: an ultrasound exploration revealed a large intravesical tissue mass. A transurethral resection of the bladder was performed, and pathological examination came back in favor of an undifferentiated round cell tumor process infiltrating the bladder muscle (Figure 1). A chest-abdomen-pelvis CT scan showed a lesional process of the bladder floor budding into the bladder lumen measuring 61 x 71 x 81 mm with micronodular infiltration of the pelvic fat without detectable adenopathy (Figure 2). A second transurethral resection of the bladder was performed the following month, with the same anatomopathological conclusion. Immunohistochemical phenotyping study was necessary, anti-P63 antibody (Clone 4A4, Bio SB), anti-synaptophysin antibody (Clone polyclonal, Bio SB), anti-PS100 antibody (Polyclonal, Dako) anti-CD99 antibody (Clone BSB-9, Bio SB), and anti-NSE antibody (Clone PBS/NC/V1/H14, DAKO) were positive. The anti-Myogenin antibody (Clone F5D, Bio SB), anti-Desmin antibody (Clone D33, Bio SB), anti-CD20 antibody (Clone L26, Bio SB), anti-CD3 antibody (Clone RBT-CD3e, Bio SB), anti-GATA-3 antibody (Clone L50-823, Bio SB), anti-Myeloperoxidase antibody (Clone Polyclonal, Bio SB), anti-CD34 antibody (Clone QBEnd/10, BIOCARE) anti-Pancytokeratin antibody (Clone AE1/AE3, Bio SB), anti-chromogranin antibody (Clone LK2H10, Bio SB), anti-EMA antibody (Clone E29, Bio SB), anti-GFAP antibody (Clone G-A-5, Bio SB), anti-neurofilament antibody (Clone 2F11, DAKO), anti-HMB45 antibody (Clone HMB45, Bio SB) and anti-H-Caldesmone antibody (Clone BSB-19, Bio SB) were all negative. The immunohistochemical profile was in favor of a round cell tumor of the Ewing sarcoma type. Molecular analysis via FISH was requested and a block of paraffin-embedded tumor tissue was sent to a specialized center in France, unfortunately the technique failed on two occasions due to transit delays related to the coronavirus pandemic. The evolution was marked by the accentuation of hematuria which motivated the realization of a new thoraco-abdomino-pelvic scanner in October, finding a tumoral mass, measuring 74 x 87 mm and infiltrating the prevesical fat by a nodule with respect of the abdominal wall as well as a moderate bilateral ureterohydronephrosis without detectable adenopathy classified pT3b N0 M0 (Figure 3). An MRI was also performed, with the same conclusion as on the CT scan (Figure 4). Bone scan did not find any secondary bone location (Figure 5).
Therapeutic intervention: the bladder resections he underwent allowed staging of the tumor and reduction of the tumor mass before initiation of chemotherapy. After 9 cycles of chemotherapy consisting of vincristine, actinomycin, cyclophosphamide, iFOSFamide, and etoposide, the patient was seen again for control with an MRI showing a clear regression of the tumor mass of about 70% (Figure 6). A resection was scheduled to reduce the tumor mass which was difficult to resect, hard and whitish. Anatomopathological examination showed no residual tumor tissue, as did molecular analysis via FISH. A final cycle of chemotherapy was scheduled.
Follow-up and outcomes: after the last cycle of chemotherapy, the patient reported an improvement in his general condition with cessation of hematuria. Radiologically and endoscopically, no tumor residue remained. The patient tolerated well all aspects of his complete management, including bladder resections and even chemotherapy, despite its side effects, which were mild for this young subject.
Patient perspective: being a minor, the patient's opinion regarding his therapeutic care was made through his father who appreciated the good multidisciplinary care and the involvement of the different parties, especially the mutual insurance company which allowed him to obtain quality care as well as free chemotherapy sessions.
Informed consent: informed consent was obtained from the father for scientific purposes and for publication.
Ewing's Sarcoma (ES) is a small round cell malignancy, very aggressive and most often of interest in bone and soft tissues such as lung, heart, pancreas, ovary, uterus, cervix, liver, rectum, parotid gland, testicles, kidney and finally bladder, of which only 24 cases have been reported in the literature. Due to the extreme rarity of this last location, no therapeutic consensus could be established and is highly inspired by the therapeutic protocol of Ewing's Sarcoma of the bone Table 1. The age of the patients ranged from 2 to 8 years, with a mean age of 41 years with no gender predominance, with 13 women and 11 men. The Asian race is the most affected, with 14 cases. The risk factors found were mainly immunosuppression; in this series, five patients were immunosuppressed (renal transplantation, chemotherapy for squamous cell carcinoma of the bladder, Hodgkin's disease, acute lymphoblastic leukemia, chemotherapy for a malignant tumor of the thigh). Hematuria and dysuria were the most prevalent symptoms. In addition, some patients had advanced disease with bilateral ureterohydronephrosis, renal failure and lower limb edema.
The paraclinical diagnosis and staging of the disease was based on ultrasound, CT scan, intravenous pyelography, exploratory cystoscopy, magnetic resonance imaging and bone scan. Five cases were metastatic at diagnosis, 3 had perivesical infiltration, and 3 had lymph node involvement. Tumor development was predominantly exophytic and occasionally endo and exophytic, with a gray and necrotic macroscopic appearance. The size of the tumor at the time of diagnosis was often large, with an average of 8 cm long. Surgical treatment was case by case, radical cystectomy was performed in 8 patients and partial resection in 5 other patients. The remaining patients were treated by transurethral resection or conservatively with palliative procedures in view of the stage of their disease. The bladder localization of Ewing's sarcoma is extremely rare, it appears like any other anatomopathological type of bladder tumor and can be confused with other malignant small round cell tumors, especially neuroendocrine small cell carcinoma, desmoplastic small round cell tumor, small cell variant of malignant melanoma, embryonal rhabdomyosarcoma, synovial sarcoma, and non-Hodgkin lymphoma.
As for the exact diagnosis, immunohistochemical study as well as molecular analysis allow deciding, the latter looks for a rearrangement of the EWSR 1 gene via FISH, and the reciprocal translocation t (11; 22) (q24; q12) and the fusion transcript EWSR1-FLI1 (85%) or EWSR1-ERG (10%) via RT-PCR. The EWSR1 gene rearrangement is also observed in Ewing-Like round cell tumors with EWS-non-ERG partner translocation, however, its fusion partner in this category is the NFATC2 or PATZ1 gene and not FLI-1 [1]. Advanced age, metastatic disease, and limited surgical attitude are associated with poor prognosis. Multidisciplinary management is required, so urologists, pathologists, oncologists and radiotherapists must be involved to optimize the patient's chances. Surgery and chemotherapy are the keywords. Surgery must be decided on a case-by-case basis depending on the extension, the impact on the upper limb and the various defects of the subject. Chemotherapy protocols based on cyclophosphamide, vincristine, actinomycin D and doxorubicin have been associated with better results [2]. The combination of ifosfamide and etoposide can also improve outcomes in non-metastatic Ewing's sarcoma/NPET, but this is not effective in metastatic disease [3,4]. Although the response of sarcomas to high-dose radiotherapy is well-defined, its use in ES/PNET of the bladder is not yet well established (4). New therapeutic approaches, including targeted therapies and immunotherapies, have recently been investigated. Combination therapies may improve efficacy and decrease resistance. Real-time kinematic (RTK) inhibitors have shown synergistic effects with chemotherapeutic agents, while mTOR pathway inhibitors have been more effective in combination with other agents, such as IGF1R inhibitors [5].
The bladder localization of Ewing's sarcoma due to its rarity requires a codified consensus during diagnosis and management due to the low survival rate. With the increasing interest of the medical profession in rare diseases and the publication of new cases and long-term follow-up results, this has become possible. Surgery combined with chemotherapy should be considered as the best option. However, the emergence of new therapies may change this situation.
The authors declare no competing interests.
All the authors have read and agreed to the final manuscript.
Table 1: comparative table of cases reported in the literature
Figure 1: microscopic aspect of the lesion: (A, B) hematoxylin and eosin x 40: round cell proliferation infiltrating the bladder wall; (C) hematoxylin and eosin x 200: higher magnification of small round cell proliferation, monomorphic in diffuse sheets; (D) anti-cytokeratin antibody AE1/AE3: negative on tumor proliferation, positive on normal urothelial surface lining; (E) anti-synaptophysin antibody: cytoplasmic positivity of tumor cells (F) anti-CD99 antibody: membrane positivity of tumor cells
Figure 2: lesional process of the bladder floor budding into the bladder lumen measuring 61x71x81mm with micronodular infiltration of pelvic fat
Figure 3: tumor mass measuring 74x87mm and infiltrating the prevesical fat and bilateral ureterohydronephrosis
Figure 4: bladder tumor process infiltrating the pre-vesical fat with bilateral ureterohydronephrosis, without obvious parietal extension
Figure 5: absence of scintigraphic signs in favor of secondary bone localization
Figure 6: clear regression of the tumor mass by 70% on control MRI
- Sbaraglia M, Righi A, Gambarotti M, Dei Tos AP. Ewing sarcoma and Ewing-like tumors. Virchows Archiv. 2020 Jan;476(1):109-119. PubMed | Google Scholar
- Esiashvili N, Goodman M, Marcus RB Jr. Changes in incidence and survival of Ewing sarcoma patients over the past 3 decades: surveillance epidemiology and end results data. J Pediatr Hematol Oncol. 2008 Jun;30(6):425-30. PubMed | Google Scholar
- Grier HE, Krailo MD, Tarbell NJ, Link MP, Fryer CJ, Pritchard DJ et al. Addition of ifosfamide and etoposide to standard chemotherapy for Ewing´s sarcoma and primitive neuroectodermal tumor of bone. N Engl J Med. 2003 Feb 20;348(8):694-701. PubMed | Google Scholar
- Senol Tonyali, Sertaç Yazici, Ergen A. The Ewing´s sarcoma family of tumors of urinary bladder: a case report and review of the literature. Balkan Med J. 2016 Jul;33(4):462-466. PubMed | Google Scholar
- Hongjiu Yu, Yonggui Ge, Lianying Guo, Lin Huang. Potential approaches to the treatment of Ewing´s sarcoma. Oncotarget. 2017;8(3):5523-5539. PubMed
Search
This article authors
On Pubmed
On Google Scholar
Citation [Download]
Navigate this article
Similar articles in
Key words
Tables and figures
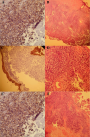 Figure 1: microscopic aspect of the lesion: (A, B) hematoxylin and eosin x 40: round cell proliferation infiltrating the bladder wall; (C) hematoxylin and eosin x 200: higher magnification of small round cell proliferation, monomorphic in diffuse sheets; (D) anti-cytokeratin antibody AE1/AE3: negative on tumor proliferation, positive on normal urothelial surface lining; (E) anti-synaptophysin antibody: cytoplasmic positivity of tumor cells (F) anti-CD99 antibody: membrane positivity of tumor cells
Figure 1: microscopic aspect of the lesion: (A, B) hematoxylin and eosin x 40: round cell proliferation infiltrating the bladder wall; (C) hematoxylin and eosin x 200: higher magnification of small round cell proliferation, monomorphic in diffuse sheets; (D) anti-cytokeratin antibody AE1/AE3: negative on tumor proliferation, positive on normal urothelial surface lining; (E) anti-synaptophysin antibody: cytoplasmic positivity of tumor cells (F) anti-CD99 antibody: membrane positivity of tumor cells