Understanding barriers to access and utilization of cervical cancer screening services among women living with HIV in Kenya and Uganda: a cross-sectional study
Katwesige Wycliff, Kathya Cordova-Pozo, Hagono Peter, Ebenezer Obi Daniel, Abul Kalam
Corresponding author: Public Health Emergencies-Uganda Red Cross Society, Kampala, Uganda| Institute for Management Research, Radboud University, The Netherlands, Department of Health Sciences, University of Groningen, University Medical Center Groningen, Groningen, The Netherlands| Hoymas Medical Center, Nairobi, Kenya| CUAMM Rumbek Centre, Lakes State, South Sudan|| Helen Keller International, Dhaka, Bangladesh 
Received: 29 Oct 2021 - Accepted: 13 Aug 2022 - Published: 29 Aug 2022
Domain: Oncology,Chronic disease prevention,Reproductive Health
Keywords: Invasive cervical cancer screening, women, HIV, health belief model, barriers, Uganda, Kenya
©Katwesige Wycliff et al. PAMJ Clinical Medicine (ISSN: 2707-2797). This is an Open Access article distributed under the terms of the Creative Commons Attribution International 4.0 License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Cite this article: Katwesige Wycliff et al. Understanding barriers to access and utilization of cervical cancer screening services among women living with HIV in Kenya and Uganda: a cross-sectional study. PAMJ Clinical Medicine. 2022;9:45. [doi: 10.11604/pamj-cm.2022.9.45.32219]
Available online at: https://www.clinical-medicine.panafrican-med-journal.com//content/article/9/45/full
Research 
Understanding barriers to access and utilization of cervical cancer screening services among women living with HIV in Kenya and Uganda: a cross-sectional study
Understanding barriers to accessibility and utilization of cervical cancer screening services among women living with HIV in Kenya and Uganda: a cross-sectional study
![]() Katwesige Wycliff1,&,
Katwesige Wycliff1,&, ![]() Kathya Cordova-Pozo2,
Kathya Cordova-Pozo2, ![]() Hagono Peter3,
Hagono Peter3, ![]() Ebenezer Obi Daniel4,
Ebenezer Obi Daniel4, ![]() Md Abul Kalam5
Md Abul Kalam5
&Corresponding author
Introduction: invasive cervical cancer (ICC) is preventable and curable. However, it is the fourth leading cause of cancer mortality among women globally, especially in low- and middle-income countries. Whereas studies reveal the existence of ICC pre-cancerous lesion screening services in Kenya and Uganda, uptake of these services remains low, highlighting the existence of unidentified barriers. This study explored factors that impede access and utilization of pre-cancerous cervical screening services among women living with HIV in low-resource settings in Uganda and Kenya.
Methods: a cross-sectional study design was used in six selected health facilities in Uganda and Kenya. A systematic cluster randomized sampling was used to select health facilities for women living with HIV who were invited to participate in the interviews. Data collection, coding, categorization, and statistical analysis of quantitative data were employed to rank and correlate among the most critical factors of the Health Belief Model framework.
Results: the results showed a negative correlation between the perceived barriers and the risk of accessing the services (-0.95, p-value 0.003) and the perceived severity (-0.95, p-value 0.004), both mean that the barriers for the women are so high that they prefer to delay the visit to the hospital. More so, there was a positive correlation between perceived risk for ICC and perceived severity (0.90, p-value 0.01), meaning that a high perception of risk of ICC will increase its severity. Health system barriers to access such as lack of supplies, information, and limited staffing were also revealed.
Conclusion: the study uncovered the health system barriers affecting ICC screening services at health facilities in Kenya and Uganda and their effects on the perceived illness. Also, it highlights the strengths and challenges while providing recommendations to promote health that includes the most vulnerable women with HIV living in socially deprived areas.
According to the World Health Organization (WHO), cervical cancer grows in the woman´s cervix with cells capable of spreading to other parts of the body, and 99% are due to high-risk human papillomaviruses (HPV) infections [1]. The Global strategy to eliminate invasive cervical cancer (ICC) includes HPV vaccination, screening, and treatment [1]. The ICC is ranked as the fourth most common cancer in women globally, accounting for approximately 570,000 new cases, with 31,000 deaths, representing 6.6% of all female cancers in 2018 [1]. This leads to a lower Human Development Index (HDI), particularly for women in low-and medium developing countries, where nearly 90% of ICC's global mortality occurs in low- and middle-resource countries, including Uganda and Kenya [2]. During the same year, Uganda (54.8%) and Kenya (33.8%) were ranked among the first 20 countries with the highest rates of ICC globally as 7th and 10th, respectively [2]. One study in Uganda indicated that HIV-positive women have a higher risk of coinfection with Human Papilloma Virus (HPV) types, HPV reactivation, and the development of ICC [3]. Also, another study in Kenya uncovered that HIV could significantly increase the risk of cervical pre-cancer to cancer development and a 6% prevalence of cervical pre-cancer and cancer among HIV-positive women [4]. Even though in Kenya and Uganda, ICC is a public health concern as it leads to morbidity and mortality among women, particularly those with HIV coinfection, the above studies did not explore context-specific and comparative factors that hinder access and utilization in the two countries.
In 2019, Kenya and Uganda were among the two countries with the third-largest epidemic of HIV in the world, each with 1.5 million people living with HIV. Besides, there was a 5% of HIV prevalence among adults (ages 15-49), and among them, only 80% were receiving antiretroviral treatment (ART) among adults [1]. A more recent study conducted in Kenya revealed that out of the 1180 women interviewed, 16.4% had been screened for ICC pre-cancerous lesions [5]. For those unscreened women, 67.9% were already aware of ICC screening. The same study also revealed high screening rates in more educated women. However, the study did not explore more reasons for this and the need for more qualitative inquiry. Besides, the uptake of ICC screening services in Kenya is deficient at 3.2%, below the target of 70%, and hence, the need to study the factors that lead to low uptake of the screening services [6]. In Uganda, one study indicated that 98.9% of women living with HIV did not think it was necessary to be screened for ICC lesions, despite being at higher risk than their HIV-negative counterparts [7]. In 2013, the Uganda Ministry of Health planned to enhance targeted interventions to improve free access to essential health services, including ICC pre-cancerous screening using Visual Inspection under Acetic Acid (VIA) [8]. In line with the above, the United States Agency for International Development (USAID) supported ICC screening programs in Kyenjojo District western Uganda through health workers' training and equipment. However, the district's uptake of these services, especially for women with HIV, remained low, yet the underlying factors remain unknown [9].
The knowledge gap was that although other studies reported that three in four HIV-positive women remained under-screened for ICC, they focused on a single country and did not compare how this affects other countries like Kenya [10]. Besides, various socio-economic, political, and cultural barriers may hinder effective HIV prevention programming in Kenya and Uganda. This implies that new HIV infections and more risk to ICC are expected to rise in coming years, with annual new HIV conditions projected to multiply around 340,500 in 2025 if no innovative interventions are implemented [11]. Astonishingly, the empirical gap was two-fold. The limited research in the study locations to explore the multi-faced barriers to access and uptake of ICC services among women with HIV in the hospitals informed the need for this study to fill the knowledge gap [5,12]. Also, limited studies in the two countries address the knowledge gap on the underlying socio-cognitive factors affecting ICC pre-cancerous via screening services. Based on the existing HBM framework and literature, our objective was to replicate, scrutinize and understand why there was limited access and utilization of the ICC screening services among women living with HIV in six selected health facilities of Kenya and Uganda. The underlying hypothesis was that the social-cognitive factors and their understanding could prompt action to increase access and utilization of the ICC screening services with the following formula: d% (uptake ICC | knowledge HBM - uptake ICC | No knowledge HBM) >0. This framework helped discover facts and empirical data on Kenya and Uganda while providing a scientific means to integrate across the medical and social sciences to improve the health of women with HIV [13]. Hence, this study was aimed at assessing the factors that hinder access and utilization of ICC pre-cancerous screening services among women with HIV in Uganda and Kenya based on the health belief model (HBM) framework [14]. The research objective was to help improve the existing policy of ICC screening by understanding why there are limited access and utilization of ICC screening services among women living with HIV in six selected health facilities of Kenya and Uganda.
Study design: a cross-sectional study design employing quantitative and qualitative techniques was employed [15]. The study's validity and reliability utilized the HBM framework in two different countries and multiple settings. The quantitative approach was two-fold. First, it tested whether the results were robust enough and unbiased. Second, it observed the main correlations among the components. The qualitative research was underpinned by ethnographic theory to understand and interpret intricate behavioural patterns [16]. Inductive and deductive (pseudo-deductive) approaches were employed based on the health belief model (HBM) theoretical framework [16,17].
Study area and period: in Uganda, the study was conducted in Kyenjojo Hospital, a public health facility in a resource-deprived setting of Kyenjojo district, Western Uganda. It has a total population of 422,204 people, whereby 39.7% of the females above 18 years are illiterate [18]. About 66% of the households are within five kilometers of proximity to the nearest public health facility [18]. In Kenya, the study was conducted at five hospitals in three different counties of Kenya, Nairobi (3.2 million population), Kiambu (2.5 million), and Kajiado (1.18 million people). The Coptic Hospital (Nairobi County) is a church-based health facility managed by the Orthodox church and is situated 4.5 km from the Nairobi Central Business District (CBD). The Kiambu hospital (Kiambu County) is located about 16 km from the Nairobi CBD. Kajiado County borders Nairobi to the South, with its headquarters about 80.5 km from Nairobi city center with three public hospitals for the study, namely, Ngong, Isinya, and Kitengela. Most residents in these two counties live in deprived-resource settings and are either peasant farmers or pastorals. The data collection process lasted three weeks (mid-January until February 2019) in Uganda and eight weeks (January until mid-March 2020) in Kenya. The goal was to collect cross-sectional data from two countries.
The study population and participants
Source population: the source population included six study locations or health facilities in Kenya and Uganda.
Study population: the study population included women attending ART services in the selected study locations.
Study units: there were five hospitals in Kenya and one hospital in Uganda.
Participants: this study involved participants coming to the study units during 2019 and 2020.
Inclusion criteria: women of reproductive age (15-49 years) and living with HIV, following antiretroviral therapy (ART), and attending sexual and reproductive health care in the included health facility.
Exclusion criteria: participants were excluded by age, meaning below 19 years and above 49 years old. Also, expert clients excluded women caretakers in ART Clinics who were HIV negative after health education sessions. Eligible women who refused to consent to participation were also excluded.
Sampling method and procedure: a systematic cluster randomized sampling was employed to select health facilities from which women living with HIV invited to participate in the interviews. First, the districts were randomly identified using the population proportion to size method. Then within each district, Hospitals were randomly selected while accounting for rural/urban locations. The number of hospitals per district varied for each country depending upon the specific sample size calculations. The selected participants were recruited from six hospitals that provide these services in Uganda and Kenya. For the study participants, the probability was proportionate to the stratum sample size with correction in the group of women with HIV in the chosen hospitals of Uganda and Kenya. To eliminate the selection bias, we used different health facilities in each country [19]. We tried to give the same potential of participation to the persons by picking Monday and Tuesdays as the days patients go the most frequently to the hospital. Participants were invited by trained research assistants who dispensed a questionnaire. To confirm the results of the questionnaires, similar participants were recruited for focus group discussions (FGDs). These sources were comparable as both used the same interview guideline structure (see data collection).
Sample size estimation this study involved a small sample. Therefore, a modified Cochran formula for samples with small populations was employed, including a ±7.5% margin of error using Table 1 [20]. Modified Cochran formula:
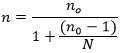
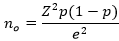
Data sources and data collection: different data collection techniques were used to capture diverse information and insights and complement each approach until reaching convergence in the most critical aspects while capturing the topic's multiple perspectives (using different moderators´ locations to reach deep saturation and comprehensiveness) [15]. This also ensured the collected data's validity and reliability of its results [15,21] and helped with triangulation which was based on the results of the different techniques and literature revision. A structured interview guideline based on the HBM model (risk perception of ICC, awareness of ICC screening programs, perceived severity, barriers that impede access to ICC, and motivators to ICC screening) with open-ended questions (divergence) was used. The content used literature, and study experts validated the pilot test. This guideline was implemented for FGDs, in-depth interviews, key-informant interviews, and questionnaires. A well-structured self-administered questionnaire was administered to consenting respondents. In the case of the uneducated or those unable to read or write, a translated questionnaire was administered verbally by an interviewer. The FGDs followed an interview guide and were carried out by a research assistant. The Research Assistants (RA) trained for two days and piloted the data collection tools to later collect data in the field using local language (Runyoro-rutooro in Uganda and Swahili in Kenya). Respondents signed informed consent before the interview or questioneer.
Data analysis: the information from open-ended questionnaires and the FGDs were transcribed verbatim by the principal and co-investigator, who also coded within the HBM themes. A deductive research approach based on the HBM involved coding, memo writing, comparison, and categorization of emerging data [20]. No software was employed for qualitative analysis. Instead, a thematic analysis was applied for qualitative data sources, and an aggregation method with scoring (per number of participants in each hospital) was used to synthesize the converging information. The main perceived aspects were explained under a rank-order per HBM component [21]. Only topics that represented 50% or more were included in the analysis and clustered in each HBM component. Every HBM component was analyzed as a variable for the quantitative analysis. The quantitative analysis involved logistic regression in creating odds ratios to determine the strength of association among independent and dependent variables using a level of significance of p < 0.05. This obtained the shares, the main descriptive statistics, and the 95% confidence interval (α=0.05). Statistical tests were applied to check for external validity and reliability [22]. Also, during a correlation test among the HBM components [21]. A normality test to check if the sample size was large enough to reflect the population is a standard probability distribution in the natural world [23]. Incomplete questionnaires were excluded from all analyses. Once the underlying factors were validated, a correlational analysis was done using SPSS (Version 27). Interaction or effect modification was measured and controlled with the control of confounding in the stratified analysis and later in the variance analysis while comparing with each HBM element for two countries [15]. The list-wise deletion option eliminated individual items missing data in SPSS v.27.
Ethics approval: the study was reviewed and registered in Uganda by the Texila American University School of Public Health Ethics committee and The Aids research Organisation Ethical Review Committee under registration number TASOREC/076/18-UG-REC-009. The University of Nairobi School of Public health's ethics and research committee also approved the study in Kenya under registration number NACOSTI/P/18/76835/200085.
Women living with HIV within reproductive ages participated (15-49). The mean age of respondents was 36.2 in Uganda and 35.4 in Kenya, with SD±7.0. Majority, 31% and 36% in Uganda and Kenya respectively, had not completed primary education. Also, 42% and 39% of women in Uganda and Kenya respectively had completed their primary education. Astonishingly, in Uganda, 57% of women were single or widowed and 27% were married. In Kenya, 66% were single or widowed and only 25% were married. Economically, most women in Uganda, 49% were peasants and 32% businesswomen. While in Kenya, 61% were businesswomen, 23% peasants. One hundred and sixty out of 146 women completed the questionnaire, with a high response rate for the calculated sample with a 7.5% margin of error. Ninety-nine women in Kenya and 24 in Uganda participated in the FGDs, which helped us capture multiple perspectives and converge in the following results. Respondents, Thirty-eight (38) sub-themes for Kenya and 61 for Uganda were categorized within the HBM framework. Table 1 and Table 1 (suite) describe the 50% or more of the data with the most mentioned sub-themes in Kenya and Uganda.
Perceived access barriers: Table 1 displays why women with HIV do not access ICC screening services. A common reason in both countries is that health workers with a lack of interest or poor cooperation hinder access to services. Other reasons are particular to each country among which are the unawareness of the ICC services, for the examination procedure (metallic instruments, the pain, and exposure), a lack of knowledge and information about ICC screening services, complications of referral (transport costs, accessing the services, time in the queue). Besides, confusing media information regarding traditional healers' capacity to cure every disease misleads towards cancer prevention.
Perceived benefits: Table 1 shows driver factors as motivators for the uptake of ICC screening services. Common drivers are free ICC screening services, trust in health services, and healthcare providers. Women that make regular visits to the facility for their HIV conditions may be offered to get the screening at no cost. Understanding the benefits of knowing their ICC status and the hope to access free treatment is crucial for screening. And the willingness of expert clients to be trained to provide health education on ICC in Uganda.
Perceived risk: Table 1 (suite) shows the main aspects that women living with HIV feel about the risk of having cervical cancer. There are no common factors among the 50% topics. Still, the ones mentioned in Kenya are that all women are at risk regardless of sexual activity, multiple sexual partners, or women with HIV who are vulnerable to ICC. In Uganda, most women with HIV believe that ICC testing was only mandatory for women who had symptoms or that poverty is a significant factor.
Perceived severity: Table 1 (suite) shows the main aspects that women living with HIV feel about cervical cancer severity (to their body). In both countries, women living with HIV coincide that cervical cancer leads to death and no cure.
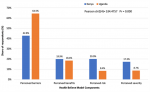
Validity, reliability, and generalizability: robustness cannot be assumed in social science because of the small sample sizes. Hence, to test the results' robustness, a normality test is a necessary condition to obtain parameter estimates that are efficient and unbiased [22]. Table 2 shows the statistical description and analysis for robustness. A skewness probability with a p-value of skewness > 0.05 for all topics except for perceived severity implies that skewness is asymptotically normally distributed. Similarly, the Kurtosis probability indicates that it is also asymptotically distributed for all subjects (p-value of kurtosis > 0.05). Finally, the joint probability chi-square of Skewness and Kurtosis is more significant than 0.05 (α = 0.05) for all topics except for the perceived severity. Subsequently, the null hypothesis cannot be rejected, which means that residuals show a normal distribution for all components except for Kenya's perceived severity. In Uganda, perceived risk and severity could not be tested due to insufficient observations. These results provide an external validity or generalizability of the products for these two countries. Figure 2 examines the relationship between the two countries' HBM components. The Chi-square value was 534.476 with a p-value below 0.05 validates the alternative hypothesis, indicating a relationship between countries and the HBM components, rejecting the null hypothesis. This figure shows that the HBM components are comparable, even though each country has its particularities, as presented above (Figure 2). For both countries, perceived barriers have the highest share (42.8% for Kenya and 64.5% for Uganda), followed by the perceived benefits or motivators (19.9% for Kenya and 18.4% for Uganda). The illness's perceived risk and severity are lower than 20% in Kenya and even lower than 9% in Uganda. This means that the factors that affect the uptake of ICC screening services among women that live with HIV in Kyenjojo District, Western Uganda, and in the three counties of Kenya (Nairobi, Kiambu, or Kajiado) were mainly based on the health system's barriers rather than in the risk or severity.
This analysis uncovered more detail by running categorical correlations among both countries' HBM components (Figure 3). Figure 3 displays the Pearson correlation coefficients, which describe the direction and strength of the linear relationship between two HBM components under the assumption of normal distribution [21,22]. This shows the validity and reliability of results for the HBM framework. There is a negative relation between the perceived barriers and the perceived risk (-0.95, p-value 0.003). This implies that if women with HIV have higher perceived barriers, their perceived risk for ICC is lower, which leads to a longer time to go to the hospital. Also, there is a negative relation between the perceived barriers and the perceived severity (-0.95, p-value 0.004). This means that if women with HIV have higher perceived barriers, the lower is their perceived severity for ICC, which leads to a longer time to go to the hospital. Finally, the perceived severity and perceived risks are positively correlated (0.90, p-value 0.01), meaning that if women with HIV have a high perceived risk of ICC, this will also increase their perceived severity.
The study was carried out among the countries with the highest rate of cervical cancer, Kenya and Uganda. The findings present the relation of the HBM components with barriers to access ICC screening services. The first is a negative relation between the perceived barriers to access the services, perceived risk for having ICC (-0.22), and perceived severity (-0.32). Consequently, this impacts the likelihood of undergoing ICC screening. The second is a positive relationship between the perceived risk for ICC and the perceived severity (+0.90). Statistical analysis showed that results are valid and reliable. This is in line with the study of Mitchell, indicating that 98.9% of women with HIV did not think it was necessary to be screened for ICC despite being at higher risk than their HIV-negative counterparts [23]. More, the uptake of ICC screening in Kenya is at 3.2%, below the targeted 70% [23,24].
Perceived barriers and the negative correlation with perceived risk and perceived severity: despite both governments' efforts to train staff and provide ICC screening infrastructure and services to vulnerable persons, there were barriers to access services (64.5% and 42.8% in Uganda and Kenya, respectively). The hindrances described included problems with the health personnel having long queues in Kenya and lack of knowledge, interest, or fear of accessing the ICC services. These findings are, to a certain extent, related to women with HIV conducted in urban settings in Uganda [25], [26], Kenya [27], and Nigeria [28], which could positively influence the validity of this research. Other studies mention that ICC screening's knowledge and awareness do not increase service uptake [29]. However, these studies focused on specific aspects, while our research is rank-ordered and correlated the most important components to present cues to action. Our study indicates that the higher the perceived barriers, the less perceived risk. Phrases like “ visiting health facilities is common only when one has developed pain or symptoms of the disease ” Before this happens, the patient has a lower perceived risk or severity for ICC. Some studies focus on the need for culture-specific, sensitive information and interventions to improve the ICC screening uptake among women with fear [25,27,29,30]. Other studies focused on monetary barriers like Zimbabwe, as many could not afford ICC screening because of its costs [30]. In theory, women who knew the underlying risks or perceived severity would get ICC screening to know their status. However, when the barriers are too high, they ignore these perceptions. In Uganda, a survey revealed that Cervical Screening uptake was deficient among HIV-infected women [9]. Also, poor treatment outcomes and death and inaccessible referral facilities that manage ICC were mentioned as barriers in Uganda [15] and Kenya [24]. This may show that the higher the barriers, the lower their perception of risk or severity until the first symptoms appear. The study reveals a positive correlation between the perceived risk to ICC and perceived severity [31]. Unfortunately, we did not find a specific study that could sustain this specific aspect more than the relationship with the barriers presented above.
Limitations: one of the limitations is that although many FGDs were organized in Uganda, this was done only in one hospital. The inclusion of more facilities could enhance the perspective. Furthermore, the HBM components were assessed as one. A split of perceived barriers or benefits in the availability, accessibility, acceptability, contact-use, and effectiveness, could bring more insights [32]. Another limitation is the statistical analysis is based on the qualitative interpretation of the HBM elements. It includes both the open-ended questionnaire results and the FDGs valued equally. Although great efforts were made to standardize in-country clusters, increased focus on survey preparation could reduce bias and confounders. A split of both results could add more insights to the discussion. A natural limitation is reaching a significant number of participants as the vulnerable population is small, and the topic is still considered taboo. Still, we think that a significant number of participants from different settings were reached to validate the study.
Conclusions: this study uncovered a myriad of sub-themes for ICC screening services at six selected health facilities in Uganda and Kenya. These factors were analysed within the HBM framework and unveiled the relations between the perceived barriers and the length for accessing and utilizing the ICC services. Also, it revealed the positive relationship between the perceived risk and severity of the ICC. The results call for an urgent need by the two countries to increase advocacy programs to improve socio-cultural and behavior change communication and awareness on cervical cancer screening among women living with HIV. Also, health system strengthening through capacity building of staff and regular equipment of screening centers can help reduce barriers to screening. Specifically, addressing the challenges in ICC prevention among vulnerable groups like women with HIV or deprived settings of the two countries will decrease cervical cancer incidence, morbidity, and mortality and address the inequities regarding access to effective cervical cancer screening and treatment. Recommendations would focus on the perceived barriers, as they are solid in these two countries and impede access to ICC screening. More efforts could be made to improve health promotion, using the ART visits for the ICC screening, including future provision for self-swabbing at home, a new recommendation from the WHO [33].
What is known about this topic
- It is already known that Uganda and Kenya have the highest rate of cervical cancer;
- The prevalence of HIV among women in the two countries is also high;
- Women with HIV are known to have an increased risk of acquiring ICC. Hence, they are highly vulnerable.
What this study adds
- The unique idea about this research is its focus on a vulnerable population, especially women living with HIV in Uganda and Kenya;
- The study also explored health system barriers to access ICC screening services by these high-risk women, while suggesting the need by governments to strengthen health systems to address them;
- The study provides a unique comparison and relationships between underlying factors impeding access to ICC screening in two East African countries with the known highest rate of cervical cancer, underpinned by the HBM.
The authors declare no competing interests.
Katwesige Wycliff (KW) conceptualized and designed the study and obtained ethical clearance in Uganda. He implemented, supervised the study, drafted the manuscript, and revised the paper for important intellectual content. Peter Hagono (PH) collected data and obtained ethical approval in Kenya. Kathya Cordova-Pozo (KCP) analyzed the data collected, ran a statistical analysis, and drafted the manuscript. Abul Kalam (AK) designed the qualitative research. Daniel Obi Ebenezer (DOE) participated in the qualitative analysis. All co-authors participated in a critical manuscript revision and approved the final version.
We highly acknowledge all stakeholders who participated in the study and assessments.
Table 1: HBM components perceived barriers and perceived benefits with the top 50% factors mentioned
Table 1 (suite): HBM components perceived risk and perceived severity with the top 50% factors mentioned
Table 2: statistical description and analysis for robustness
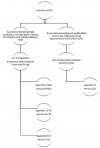
Figure 1: a flow diagram showing sampling procedures for quantitative and qualitative choices
Figure 2: relationship between HBM components in Kenya and Uganda, Pearson test of independence
Figure 3: correlations among HBM components, Kenya and Uganda
- World Health Organisation. Comprehensive cervical cancer control: a guide to essential practice 2nd edition. Geneva: WHO Library. 2014. Accessed January 15, 2019.
- Davis V, Njama-meya D, Lim J, Murray M, and Sanjose S. Opportunities and challenges for introducing HPV testing for cervical cancer screening in sub-Saharan Africa. Prev Med (Baltim). 2018;114(July):205-8. PubMed | Google Scholar
- Lin LL, Lakomy DS, Chiao EY, Strother RM, Wirth M, Cesarman E et al. Clinical trials for treatment and prevention of HIV-associated malignancies in sub-Saharan Africa: building capacity and overcoming barriers. J Glob Oncol. 2020 Jul;6:1134-1146. PubMed | Google Scholar
- Ononogbu U, Almujtaba M, Modibbo F, Lawal I, Offiong R, Olaniyan O et al. Cervical cancer risk factors among HIV-infected Nigerian women. BMC Public Health. 2013;13:582. PubMed | Google Scholar
- Nyangasi M, Nkonge NG, Gathitu E, Kibachio J, Gichangi P, Wamai RG. Predictors of cervical cancer screening among Kenyan women: results of a nested case-control study in a nationally representative survey. BMC Public Health. 2018 Nov 7;18(Suppl 3):1221. PubMed | Google Scholar
- Munoru F, Gitonga L, Muraya M. Integration of cervical cancer screening services in the routine examinations offered in the Kenyan health facilities: a systematic review. Open J Obstet Gynecol. 2019;09(05):656-68. Google Scholar
- Mitchell SM, Pedersen HN, Stime EE, Sekikubo M, Moses E, Mwesigwa D. Self-collection-based HPV testing for cervical cancer screening among women living with HIV in Uganda?: a descriptive analysis of knowledge, intentions to screen and factors associated with HPV positivity. BMC Women's Health. 2017 Jan 13;17(1):4. PubMed | Google Scholar
- USAID. STRIDES for family health legacy series: strengthening health services with private sector support in Uganda. Accessed January 15, 2019.
- Wanyenze RK, Bwanika JB, Beyeza-Kashesya J, Mugerwa S, Arinaitwe J, Matovu JKB et al. Uptake and correlates of cervical cancer screening among HIV-infected women attending HIV care in Uganda. 2017;10(1):1380361. PubMed | Google Scholar
- Craig P, Gibson M, Campbell M, Popham F, Katikireddi SV. Making the most of natural experiments?: what can studies of the withdrawal of public health interventions offer. Prev Med (Baltim). 2018;108(October 2017):17-22. PubMed | Google Scholar
- Vithalani J, Herreros-Villanueva M, Vithalani J. HIV Epidemiology in Uganda: a survey based on age, gender, number of sexual partners, and frequency of testing. BMC. 2018;18(3):523-30. PubMed | Google Scholar
- Mukama T, Ndejjo R, Musabyimana A, Halage AA, Musoke D. Women´s knowledge and attitudes towards cervical cancer prevention: a cross-sectional study in Eastern Uganda. BMC Women's Health. 2017;17(1):1-8. PubMed | Google Scholar
- Vogl S, Schmidt EM, Zartler U. Triangulating perspectives: ontology and epistemology in analyzing qualitative multiple perspective interviews. Int J Soc Res Methodol. 2019;22(6):611-24. Google Scholar
- Abraham C, Sheeran P. The health belief model. In: Cambridge handbook of psychology, health, and medicine, second edition. 2014;97-102.
- Field A. Discovering statistics using SPSS; SAGE Publications: Thousand Oaks, CA, USA, 2009;58:303.
- Bricki N, Green, J. A guide to using qualitative research methodology. Med Sans Fronteers. 2007;11-3. Google Scholar
- Leung L. Validity, reliability, and generalizability in qualitative research. J Fam Med Prim Care. 2015 Jul-Sep;4(3):324-7. PubMed | Google Scholar
- KNBS. Population by sex and age groups - KNBS Open Data Portal. Kenya National Bureau of Statistics. 2020.
- Pourhoseingholi MA, Baghestani AR, Vahedi M. How to control confounding effects by statistical analysis. Gastroenterology and Hepatology from bed to the bench. 2012;5(2):79-83. PubMed | Google Scholar
- Bartlett J, Kotrlik JW, Higgins CC. Organizational research: determining appropriate sample size in survey research. 2001. Google Scholar
- Leslie R Odom, James R Morrow Jr. What´s this R? a correlational approach to explaining validity, reliability, and objectivity coefficients. 10, Measurement in Physical Education and Exercise Science. 2006;10:137-45. Google Scholar
- Ndejjo R, Mukama T, Musabyimana A, Musoke D. Uptake of cervical cancer screening and associated factors among women in rural Uganda: a cross-sectional study. PLoS One. 2016;11(2):1-13. PubMed | Google Scholar
- Munoru F, Gitonga L, Muraya M. Integration of cervical cancer screening services in the routine examinations offered in the Kenyan health facilities: a systematic review. Open Journal of Obstetrics and Gynecology. 2019;9:656-668. PubMed | Google Scholar
- Rosser JI, Njoroge B, Huchko MJ. Knowledge about cervical cancer screening and risk perception among women attending outpatient clinics in rural Kenya. Int J Gynecol Obstet. 2015;128(3):211-5. PubMed | Google Scholar
- Modibbo FI, Dareng E, Bamisaye P, Jedy-Agba E, Adewole A, Oyeneyin L et al. Qualitative study of barriers to cervical cancer screening among Nigerian women. BMJ Open. 2016;6(1). PubMed | Google Scholar
- Hasahya OT, Berggren V, Sematimba D, Nabirye RC, Kumakech E Olivia. A qualitative study among women in Uganda following completion of an HPV vaccination campaign. Glob Health Action. 2016 Feb 16;9:29336. PubMed | Google Scholar
- Ndejjo R, Mukama T, Kiguli J, Musoke D. Knowledge, facilitators and barriers to cervical cancer screening among women in Uganda: a qualitative study. BMJ Open. 2017;7(6):1-8. PubMed | Google Scholar
- Hasahya OT, Berggren V, Sematimba D, Nabirye RC, Kumakech E. Beliefs, perceptions and health-seeking behaviors in relation to cervical cancer?: a qualitative study among women in Uganda following completion of an HPV vaccination campaign. 2016;1:1-9. PubMed | Google Scholar
- Oketch SY, Kwena Z, Choi Y, Adewumi K, Moghadassi M, Bukusi EA et al. Perspectives of women participating in a cervical cancer screening campaign with community-based HPV self-sampling in rural western Kenya: a qualitative study. BMC Women's Health. 2019;19(1):1-10. PubMed | Google Scholar
- WHO Regional Office for Africa. Assessment of barriers to accessing health services for disadvantaged adolescents in Nigeria. 2019. Accessed January 15, 2019.
- Kim HY. Statistical notes for clinical researchers: assessing normal distribution (2) using skewness and kurtosis. Restor Dent Endod. 2013;38(1):52. PubMed | Google Scholar
- Bukirwa A, Mutyoba JN, Mukasa BN, Karamagi Y, Odiit M, Kawuma E et al. Motivations and barriers to cervical cancer screening among HIV-infected women in HIV care: a qualitative study. BMC Women's Health. 2015 Oct 12;15:82. PubMed | Google Scholar
- World Health Orgarnisation. WHO guideline for screening and treatment of cervical pre-cancer lesions for cervical cancer prevention, 2nd edition. Geneva. WHO Library. 2021. Accessed January 15, 2019.