Disseminated tuberculosis with splenic and pericardial involvement on HIV-infected inpatient: ultrasound evaluation and follow-up of response to treatment (case report)
Bartolomeu Chongo, Maria Elisa Verdú, Artur Macuacua, Rosa Juisse, Edy Nacarapa
Corresponding author: Bartolomeu Chongo, Chókwè Carmelo Hospital, Department of Medicine, Daughters of Charity, Saint Vincent of Paul, Gaza Province, Mozambique 
Received: 18 Dec 2021 - Accepted: 30 May 2022 - Published: 06 Jun 2022
Domain: Internal medicine
Keywords: Disseminated tuberculosis, tuberculous pericarditis spleen, ultrasound, case report
©Bartolomeu Chongo et al. PAMJ Clinical Medicine (ISSN: 2707-2797). This is an Open Access article distributed under the terms of the Creative Commons Attribution International 4.0 License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Cite this article: Bartolomeu Chongo et al. Disseminated tuberculosis with splenic and pericardial involvement on HIV-infected inpatient: ultrasound evaluation and follow-up of response to treatment (case report). PAMJ Clinical Medicine. 2022;9:6. [doi: 10.11604/pamj-cm.2022.9.6.32869]
Available online at: https://www.clinical-medicine.panafrican-med-journal.com//content/article/9/6/full
Case report 
Disseminated tuberculosis with splenic and pericardial involvement on HIV-infected inpatient: ultrasound evaluation and follow-up of response to treatment (case report)
Disseminated tuberculosis with splenic and pericardial involvement on HIV-infected inpatient: ultrasound evaluation and follow-up of response to treatment (case report)
![]() Bartolomeu Chongo1,&, Maria Elisa Verdú1,
Bartolomeu Chongo1,&, Maria Elisa Verdú1, ![]() Artur Macuacua1, Rosa Juisse1, Edy Nacarapa1
Artur Macuacua1, Rosa Juisse1, Edy Nacarapa1
&Corresponding author
Miliary/disseminated tuberculosis is a manifestation associated with immunosuppression less diagnosed, accounting for less than 2% of tuberculosis cases. The objective of this case report is to present the case of miliary/disseminated tuberculosis in inpatient new diagnosed for HIV with severe immunosuppression in a rural area with a high prevalence of HIV/TB and the impact of ultrasound on diagnosis and follow-up. This case describes a 28-year-old male new diagnosed for HIV with severe immunosuppression, miliary pattern in chest X-ray, GeneXpert sputum detected medium, pericardial effusion and microlesions in spleen in ultrasound evaluation. Diagnosis and treatment of disseminated tuberculosis represents a challenge in a rural areas and if not opportunely diagnosed and treated in time it is inevitably fatal. Routine tuberculosis screening exams associated with image exam like ultrasound represents advancement in diagnosis and early start of treatment and follow-up.
Tuberculosis (TB) remains a major public health issue and a leading cause of death in Mozambique. In 2018, an estimated 162 000 people (551 cases per 100 000 people) fell ill with TB in Mozambique, 58 000 (36%) of whom were coinfected with TB and HIV [1]. Disseminated/miliary tuberculosis (TB) is defined as having two or more non-contiguous sites resulting from lymphohematogenous dissemination of Mycobacterium tuberculosis or infiltrates on chest X-ray were classified as typical (multiple 1-3 mm well-defined nodules throughout all lung fields) or atypical (predominant nodules that measured >3 mm or reticulonodular pattern) miliary pattern [2,3]. Disseminated TB occurs because of the inadequacy of host defenses in containing the infection, allowing the organism to proliferate and disseminate throughout the body. The emergence of the HIV/AIDS pandemic and widespread use of immunosuppressive drugs has changed the epidemiology of miliary tuberculosis [4]. The epidemiology of disseminated/miliary TB continues to determine worldwide, but in various clinical studies among immunocompetent adults, miliary TB accounts for less than 2% of all cases of TB and up to 20% of all extra-pulmonary TB (EPTB) cases. In late HIV infection, EPTB accounts for more than 50% of all cases of TB [5].
In the advanced HIV settings, the diagnosis of TB may be more difficult due to atypical clinical presentations, undiagnosed HIV infection, and disease involvement of occult sites, where sputum testing is often negative for mycobacteria [6]. Focused Assessment with Sonography for HIV and Tuberculosis (FASH) has been applied to detect signs of extrapulmonary tuberculosis, namely including pleural and pericardial effusion, enlarged abdominal lymph nodes, hypoechogenic lesions in the spleen and the liver, ascites, and thickening of the bowel wall [7]. Patients with miliary TB must be promptly treated with standard anti-tuberculosis treatment, as the disease is uniformly fatal if not treated [8]. Management of coinfected patients can be difficult due to overlapping drug toxicities and interactions. On the other hand, the initiation of antiretroviral therapy after starting TB treatment is highly recommended in HIV infected patients [9]. This report describes the case of miliary/disseminated tuberculosis in inpatient new diagnosed for HIV with severe immunosuppression in a rural area with a high prevalence of HIV/TB and the impact of ultrasound on diagnosis and follow-up.
Information for the patient: A 28-year-old patient, male, new diagnosed for HIV without ART, referred from the Rural Hospital in Chókwè within 2 months of the start of symptoms with productive cough with yellowish sputum, chest pain, recurrent fever predominantly in the afternoon, night sweats, anorexia, and progressive weight loss.. Two weeks before admission, it worsened with cough with hemoptoic sputum, worsening chest pain, dyspnea, abdominal distension and discomfort, associated with bilateral lower limb edema. The patient did not report headache, diarrhea, vomiting.
Clinical results: on physical examination on admission, in poor general condition, with dyspnea, tachycardia, anicteric, feverless, with a temperature of 35.9°C. Blood pressure was 119/70 mmhg and he had a heart rate of 101 beats per min and respiratory rate of 28 breaths per min, oxygen saturation 90% to ambient air. Weight 51.3 kg. He had slightly jugular venous distension 450 and chest auscultation revealed a bilateral diffuse fern. Cardiac auscultation light hypophonetic heart tones, rhythmic. His present abdomen is slightly distended, pain on palpation in the upper left quadrant, with a grade 1 splenomegaly. Genitals with edema, with tortuosity of the penis. Lower limbs bright, with bilateral edema Godet grade +.
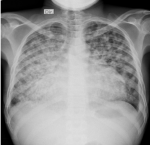
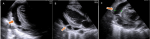
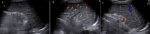
Diagnostic approach: Abnormal laboratory parameters with anemia, elevated erythrocyte sedimentation rate, low CD 4 count, and high viral load are summarized in Table 1. Chest X-ray with miliary pattern and increased cardiothoracic index (Figure 1). Transthoracic echocardiography revealed pericardial effusion with pericardial strain and fibrin (Figure 2). Transabdominal ultrasound revealed splenomegaly, echotexture heterogeneous with multiple diffuse hypoechoic micronodulares lesions (parenchymal necrosis), and Doppler echo with good parenchymal perfusion (Figure 3).
Therapeutic intervention and follow-up: we started empirical treatment before the laboratory proof with combination of 4DFC (Rifampicin, Isoniazid, Ethambutol and Pyrazinamide for two months and Rifampicin, Isoniazid for four months depending on the evolution), corticotherapy (Prednisolone 60 mg per day for six weeks), Co-trimoxazole 960 mg day, Pyridoxine 50 mg day, high protein diet. Bed 450 and rest. We prefer expectant treatment. On 12th day, the patient progress showed with satisfactory clinical and laboratory improvements, although continued with occasional cough with bloodiness sputum, no dyspnea, reduction of abdominal distension and lower limb edema (godet +), desfervescence, improved appetite and general condition. On the 14th day, he had little pericardial effusion in the ultrasound control and improvement in splenic lesions compared to admission. Chest X-ray revealed with significant improvement, mainly particularly in the left hemithorax (Figure 4). We started antiretroviral therapy (ARV) treatment on the 14th day. Patient with negative bacilloscopy on 20th day of anti-TB treatment. On day 30 was discharged to continue follow up (6 months treatment course) in the close health center. We examined the patient at the end of treatment, and he had completely recovered (Figure 5).
Informed consent: the patient agreed to publish the case for study and learning purposes, to use of any image and he was guaranteed the concealment of his identity.
The HIV pandemic altered the conception that TB is a disease of the lungs, for the new disseminated and multiorgan involvement outlook. Pulmonary and extra-pulmonary Tuberculosis is more prevalent in HIV patient who presented with lower CD4 cell counts <200 cells/mm3. In study from USA also found only 38% of TB in the lungs alone, while 30% entirely extra-pulmonary and 32% both pulmonary and extrapulmonary [10]. Disseminated TB is more common than it is diagnosed in advanced HIV disease. Miliary Tuberculosis it´s a standard pulmonary form of disseminated TB. Primarily, the disease involves the lungs and, at times, distant blood-borne spread results in the development of extrapulmonary tuberculosis. Miliary pattern on chest radiograph is considered to be the hallmark of miliary tuberculosis [11]. GeneXpert is World Health Organization-endorsed technology representing the gold standard for TB testing despite attaining less sensitivity for HIV and TB co-infected patients as compared to HIV-negative patients [12].
Imaging modalities such as conventional radiography, ultrasonography, computed tomography (CT), magnetic resonance imaging (MRI), positron emission tomography CT (PET-CT) have been used to localize the disease, assess the extent of organ involvement and evaluate response to treatment [13]. Common manifestations of EPTB include pericardial effusion, pleural effusion, and abdominal TB. Tuberculous pericarditis is consistently reported as the predominant cause of pericardial effusion in Southern Africa and is in most of the cases associated with HIV coinfection. Pericardial effusions usually present as an anechoic rim around the heart and microabscesses in the spleen present as small (between 0.5 and 1 cm), round, hypoechoic lesions in the parenchyma [14]. In adults, pericardial effusion is commonly classified according to the size including trivial (seen only in systole), mild (<10 mm), moderate (10-20 mm), and severe (>20 mm). Nevertheless, echocardiography still remains the imaging modality of choice to assess the pericardium due to its ease of use, availability, cost-effectiveness and its comprehensive appraisal of the heart and its hemodynamics [15].
The predominant symptoms of tuberculous pericarditis are cough, dyspnea, chest pain, night sweats, orthopnea, weight loss, and ankle edema are also common. As for signs, the most frequent are cardiomegaly, pericardial rub, fever, and tachycardia. Other findings may include pulsus paradoxicus, hepatomegaly, distended neck veins, pleural effusion, and distant heart tones [16]. The clinical presentation of splenic abscess is often non-specific, making the diagnosis difficult. Splenic abscess should be considered in patients presenting with fever of undetermined origin, abdominal pain and splenomegaly [17]. Our patient presented with board of symptoms and signs who include the complaints about the three organs involved (cough, hemoptysis, dyspnea, chest pain, slightly distended neck veins, abdominal pain in left the upper quadrant, splenomegaly and lower limb edema) and constitutional symptoms (fever, anorexia, weight loss and asthenia). We used the term miliary and disseminated as the same. We confirm the diagnosis of tuberculosis with Genexpert ultra of the sputum detected medium without resistance for Rifampicin. Clinical suspicion of multiorgan involvement is required to determine the true extent of the disease. Many cases improve with treatment without detected multiorgan involvement. The signs of lung disease (miliary pattern in chest X-ray) with increased cardiothoracic index associated HIV positive status with severe immunosuppression (CD4 50 cells/ul) lead for suspicion of involvement of multiorgans and disseminated disease. Echocardiography and abdominal ultrasound were conducted for the real extent of disseminated disease with pericardial effusion and spleen with suggestive features. We didn´t perform pericardiocentesis for diagnosis and therapeutic purposes or spleen aspiration. We centralized the approach in the Anti-TB drugs, based in the principle that when correct agent, correct treatment, the response is good.
Treatment of TB is curative regardless of site, if it is instituted early and if the organism remains sensitive to all first-line anti-tuberculous drugs [18]. For susceptible organisms, treatment of disseminated TB includes the administration of the “four-drug regimen,” which consists of two phases, Rifampicin, Isoniazid (INH), pyrazinamide, and ethambutol/streptomycin given daily for the first 2 months. The treatment is then continued with Rifampicin and Isoniazid for a further 4 months, with the possibility of being extended to 7 months in some cases. In general, adjunct corticosteroid treatment can be given in disseminated TB with meningitis, pericarditis, and adrenal insufficiency and in disseminated TB with refractory hypoxemia [19]. The World Health Organization (WHO) recommends dolutegravir (DTG)-based antiretroviral therapy (ART) as a preferred first-line regimen for PLWH initiating ART. Dolutegravir produce rapid virologic and immunologic responses in HIV treatment-naive patients who have drug-susceptible TB and are taking Rifampicin-containing TB treatments, provided the DTG dose is increased to 50 mg twice daily during (and for 2 weeks after) TB treatment. Among HIV treatment-naive individuals, antiretroviral therapy (ART) should be started within 2 weeks of TB treatment initiation for patients with a CD4+ lymphocyte count less than 50 cells/mm3 and within 8 weeks for those with higher CD4+ values. Cotreatment, while reducing risk of death and new opportunistic infections, poses challenges, owing to overlapping toxicities, drug interactions, and immune reconstitution inflammatory syndrome (IRIS) [20].
We began treatment as soon as possible with the first line anti-tuberculosis drugs based on the clinical symptoms before the laboratory confirmation with a combination of Isoniazid, Rifampicin, Pyrazinamide, and Ethambutol for the first 2 months and isoniazid and rifampicin for 4-7 months depending on the response to treatment. Associated treatment was based in Adjunct Corticosteroid, Cotrimoxazole to prevent related opportunistic infections, pyridoxine to prevent peripheral neuropathy. Patient recommended to rest in the bed 45°. We didn´t consider meningeal and joint involvement, being objective when it presents, and our patient didn´t refer neurological and skeleton complaints. We began ARV treatment on the 14 th dayTB treatment, with DTG boost as soon as recommended limiting adverse events related to HIV infection, and opportunistic disease and allowing time to control the mycobacteria infection first to minimize risk for Immune reconstitution inflammatory syndrome (IRIS). We used an integrated approach for treatment of tuberculosis, based on the anti-TB treatment in is large features, associated diseases (HIV) and related associated manifestations (undernutrition). Considering the malnutrition status, the diet was based on hyper protein and caloric diet. We did an ultrasound to control the response to treatment on the 14th day and at the end of treatment. The limitation in this work was the scarcity of data about miliary and disseminated tuberculosis in Mozambique, little training in lung ultrasound and the institutional inability to perform histopathological exams. We recommend the use of ultrasound as part of the algorithm in tracking, diagnosis, and follow-up the response to tuberculosis treatment in HIV patients with severe immunosuppression.
Disseminated TB is life-threatening disease common, but less diagnosed in high burden TB/HIV settings in patient with severe immunosuppression. Disseminated disease is common among HIV patients. Routine tuberculosis diagnosis exams associated with imaging technics for diagnosis of extrapulmonary dissemination is useful. Ultrasound represents useful approach and sensitive for tracking, diagnosis and follow-up for response to treatment for disseminated (multiorgan) tuberculosis in HIV patients with severe immunosuppression. The early start of treatment with anti-tuberculosis drugs is fundamental, because it left untreated or delayed, the risk to life is potentially high.
The authors declare no competing interests.
Cenceptualization, definition of intellectual content and writing original draft: Bartolomeu Chongo. Literature search: Bartolomeu Chongo, Rosa Juisse. Supervision: Bartolomeu Chongo, Maria Elisa Verdú, Artur Macuacua and Rosa Juisse. Data acquisition: Bartolomeu Chongo, Artur Macuacua. Validation: Bartolomeu Chongo, Edy Nacarapa. Writing - review editing: Bartolomeu Chongo, Artur Macuacua, Edy Nacarapa. All the authors have read and agreed to the final manuscript.
We would like to thank Dr. Fernando De Juan Burgeño for his contribution to the discussion and interpretation of ultrasound images; and Dra Lucia Chambal for her contribution to the therapeutic approach comments.
Table 1: analysis results at admission up to 12 days of hospitalization
Figure 1: chest X-ray (PA) showing pulmonary infiltrate with confluent diffuse micronodular pattern and increased cardiothoracic index
Figure 2: A) transthoracic parasternal long axis (two-dimensional) view showing large echo-free space between the two layers of the pericardium (large pericardial effusion); B) fibrin strains in the two dimensional parasternal view C) pericardial effusion with spill column 26.3 mm
Figure 3: A) transabdominal view showing enlargement of the spleen with 11.78 cm length; B) heterogeneous echotexture with diffuse hypoechoic micronodular lesions (parenchymal necrosis) approximately 5 mm in diameter; C) splenic Doppler with good parenchymal perfusion
Figure 4: evolution (on 14 days of treatment): A) significant radiological improvement, with reduced infiltrate mainly in the left lung field, decreased cardiothoracic index; B) Transthoracic parasternal long axis (two-dimensional) view showing a decrease in the quantity of effusion; C) Transthoracic parasternal long axis (M-MODE) view showing a decrease in the quantity of effusion
Figure 5: evolution (end of treatment): A) transthoracic parasternal long axis (two-dimensional) view showing no pericardial effusion; B) transthoracic parasternal long axis (M-MODE) view showing no pericardial effusion; C) reduction of splenic length (10.12 cm) and parenchyma with no hypoechoic lesions
- Geneva: World Health Organization. Global tuberculosis report 2019. 2019. Accessed September 24; 2021.
- Ayaslioglu E, Basar H, Duruyurek N. Disseminated tuberculosis with lymphatic, splenic and scrotal abscesses: a case report. Cases J. 2009;2:6995. PubMed | Google Scholar
- Ayaslioglu E, Basar H, Duruyurek N, Kalpaklioglu F, Gocmen S, Erturk A et al. Miliary tuberculosis: epidemiologicaland clinical analysis of large-case series from moderate to low tuberculosis endemic Country. Medicine (Baltimore). 2017;96(5):e5875. PubMed | Google Scholar
- Lau A, Ferrara G. Disseminated TB: still being missed and misunderstood. Int J Tuberc Lung Dis. 2021;25(4):255-257. PubMed | Google Scholar
- Sharma SK, Mohan A, Sharma A. Miliary tuberculosis: a new look at an old foe. J Clin Tuberc Other Mycobact Dis. 2016;3:13-27. PubMed | Google Scholar
- Mbanjumucyo G, Henwood PC. Focused assessment with sonography for HIV-associated tuberculosis (FASH) case series from a Rwandan district hospital. Afr J Emerg Med. 2016;6(4):198-201. PubMed | Google Scholar
- Ndege R, Ngome O, Bani F, Temba Y, Wilson H, Vanobberghen F et al. Ultrasound in managing extrapulmonary tuberculosis: a randomized controlled two-center study. BMC Infect Dis. 2020;20(1):349. PubMed | Google Scholar
- Sharma SK, Mohan A, Sharma A. Challenges in the diagnosis & treatment of miliary tuberculosis. Indian J Med Res. 2012;135(5):703:30. PubMed | Google Scholar
- Gurkan C, Sekerbey H, Babalik A. Disseminated tuberculosis: a 3-year case-series experience in a tertiary care center. Turk Thorac J. 2021;22(5):358-363. PubMed | Google Scholar
- Takhar RP, Mirdha K, Purohit G, Maan L, Bainara MK. Impact of HIV co-Infection on clinical presentation in patients with TB and correlation of the findings with level of immune suppression. Tanaffos. 2018;17(3):188-197. PubMed | Google Scholar
- Sharma SK, Mohan A, Sharma A, Mitra DK. Miliary tuberculosis: new insights into an old disease. Lancet Infect Dis. 2005;5(7):415-430. PubMed | Google Scholar
- Saeed M, Hussain S, Riaz S, Rasheed F, Ahmad M, Iram S et al. GeneXpert Technology for the diagnosis of HIV-associated tuberculosis: Is scale-up worth it. Open Life Sci. 2020;15(1):458-465. PubMed | Google Scholar
- Sharma SK, Mohan A. Tuberculosis: from an incurable scourge to a curable disease - journey over a millennium. Indian J Med Res. 2013;137(3):455-493. PubMed | Google Scholar
- Heller T, Wallrauch C, Goblirsch S, Brunetti E. Focused assessment with sonography for HIV-associated tuberculosis (FASH): a short protocol and a pictorial review. Crit Ultrasound J. 2012;4(1):21. PubMed | Google Scholar
- Pérez-Casares A, Cesar S, Brunet-Garcia L, Sanchez-de-Toledo J. Echocardiographic evaluation of pericardial effusion and cardiac tamponade. Front Pediatr. 2017;5:79. PubMed | Google Scholar
- Trautner BW, Darouiche RO. Tuberculous pericarditis: optimal diagnosis and management. Clin Infect Dis. 2001;33(7):954-961. PubMed | Google Scholar
- Gupta PP, Fotedar S, Agarwal D, Sansanwal P. Tuberculosis of spleen presenting with pyrexia of unknown origin in a non-immunocompromised woman. Lung India. 2008;25(1):22-24. PubMed | Google Scholar
- Fanning A. Tuberculosis: extrapulmonary disease. CMAJ. 1999;160(11):1597-1603. PubMed | Google Scholar
- Khan FY. Review of literature on disseminated tuberculosis with emphasis on the focused diagnostic workup. J Family Community Med. 2019; 26(2):83-91. PubMed | Google Scholar
- Dooley KE, Kaplan R, Mwelase N, Grinsztejn B, Ticona E, Lacerda M et al. Dolutegravir-based antiretroviral therapy for patients coinfected with tuberculosis and human immunodeficiency virus: a multicenter, noncomparative, open-label, randomized trial. Clin Infect Dis. 2020;70(4):549-556. PubMed | Google Scholar
Search
This article authors
On Pubmed
On Google Scholar
Citation [Download]
Navigate this article
Similar articles in
Key words
Tables and figures
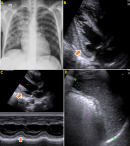 Figure 4: evolution (on 14 days of treatment): A) significant radiological improvement, with reduced infiltrate mainly in the left lung field, decreased cardiothoracic index; B) Transthoracic parasternal long axis (two-dimensional) view showing a decrease in the quantity of effusion; C) Transthoracic parasternal long axis (M-MODE) view showing a decrease in the quantity of effusion
Figure 4: evolution (on 14 days of treatment): A) significant radiological improvement, with reduced infiltrate mainly in the left lung field, decreased cardiothoracic index; B) Transthoracic parasternal long axis (two-dimensional) view showing a decrease in the quantity of effusion; C) Transthoracic parasternal long axis (M-MODE) view showing a decrease in the quantity of effusion
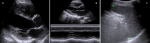 Figure 5: evolution (end of treatment): A) transthoracic parasternal long axis (two-dimensional) view showing no pericardial effusion; B) transthoracic parasternal long axis (M-MODE) view showing no pericardial effusion; C) reduction of splenic length (10.12 cm) and parenchyma with no hypoechoic lesions
Figure 5: evolution (end of treatment): A) transthoracic parasternal long axis (two-dimensional) view showing no pericardial effusion; B) transthoracic parasternal long axis (M-MODE) view showing no pericardial effusion; C) reduction of splenic length (10.12 cm) and parenchyma with no hypoechoic lesions