Determinants of anaemia among children aged 6 to 59 months in the Republic of Guinea
Gerard Christian Kuotu, Almamy Amara Toure, Aboubacar Sidiki Magassouba, Abdoulaye Doumbouyah
Corresponding author: Gerard Christian Kuotu, Faculty of Health sciences and Technology, University of Gamal Abdel Nasser of Conakry, Dixinn, Guinea 
Received: 14 Feb 2022 - Accepted: 25 Feb 2023 - Published: 28 Feb 2023
Domain: Biostatistics,Non-Communicable diseases epidemiology,Internal medicine
Keywords: Anaemia, malnutrition, "demographic and health survey", Guinea
©Gerard Christian Kuotu et al. PAMJ Clinical Medicine (ISSN: 2707-2797). This is an Open Access article distributed under the terms of the Creative Commons Attribution International 4.0 License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Cite this article: Gerard Christian Kuotu et al. Determinants of anaemia among children aged 6 to 59 months in the Republic of Guinea. PAMJ Clinical Medicine. 2023;11:46. [doi: 10.11604/pamj-cm.2023.11.46.33760]
Available online at: https://www.clinical-medicine.panafrican-med-journal.com//content/article/11/46/full
Determinants of anaemia among children aged 6 to 59 months in the Republic of Guinea
![]() Gerard Christian Kuotu1,&,
Gerard Christian Kuotu1,&, ![]() Almamy Amara Toure2, Aboubacar Sidiki Magassouba3, Abdoulaye Doumbouyah2
Almamy Amara Toure2, Aboubacar Sidiki Magassouba3, Abdoulaye Doumbouyah2
&Corresponding author
Introduction: anaemia in children aged 6 to 59 months is a major health problem in Guinea. Knowing the profile, the demographic and economic social determinants that explain the onset of anaemia could allow health policies to better target this particularly at-risk age group.
Methods: we conducted a cross-sectional study using data from Guinea's fifth Demographic and Health Survey of 2018. A multiple correspondence analysis (MCA) was performed to determine the anaemic profile of children followed by an ordinal logistic regression model with the level of anaemia considered as anaemia considerate as reference with four ordered modalities (normal, mild, moderate, and severe), to identify the determinants factors of anaemia.
Results: three (03) clusters of anaemic children have been identified: The first one with the specific features of the absence of the use of an impregnated mosquito net and the presence of mild anaemia in the mother. The second one is made up of mothers living mainly in urban areas, and the third one is made up of mothers under the age of 18. From these profiles, eleven (11) factors were identified as associated with anaemia in these children.
Conclusion: therefore, control efforts must gear towards the establishment of appropriate policies emphasizing the education of young women and geographical balance by by taking more care among children leaving in the urban areas.
Anaemia is a serious global public health problem that particularly affects young children and pregnant women. World Health Organization estimates that 42% of children less than 5 years of age and 40% of pregnant women worldwide are anaemic. In Guinea, since 2005, the prevalence of anaemia in children aged 6-59 months has not changed significantly, from 77% in 2005 [1] to 75% in 2018 [2]. In almost a third of cases (31%), children have mild anaemia; in nearly four in ten children (42%), it is moderate anaemia, and 2% suffer from severe anaemia [3]. Despite the strategies put in place by health policies to reach this group of children, only 4% received a minimum acceptable dietary intake, as recommended by the WHO, in terms of minimum dietary diversity (16%) and frequency. Minimum food intake (24%) [4]. Additionally, according to this survey, among children under 5 years of age, 30% were stunted, 9% were wasted or acutely malnourished, and 16% were underweight [5].
In the recent literature, iron deficiency anaemia is one of the main factors contributing to the global anaemia burden of the disease in areas with high endemicity of parasitic infections, such as Guinea. It is believed to be linked to low consumption and absorption of iron from food [6,7]. Intestinal parasites such as hookworm, roundworm, and schistosomiasis, acute and chronic infections including malaria, cancer, tuberculosis, and HIV may also lower blood hemoglobin levels [8,9]. However, also, the presence of other micronutrient deficiencies, including vitamins A and B12, folate, riboflavin, and copper, can increase the risk of anaemia [10,11-14]. Moreover, the household's standard of living also influences the prevalence of anaemia, which rose from 57% among women living in the poorest households to 50% among those belonging to the richest households in 2005 [1]. Consumption of iron-rich foods is essential for preventing and controlling iron deficiency anaemia [15-18]. It is also important to avoid consuming iron absorption inhibitors such as tea and coffee on meals, although this is part of the habits observed in the sub-Saharan region from which Guinea originates [19]. In addition to these other factors, there is a lack of consumption of foods of animal origin, such as red meat, which contain the highest amounts of iron and are more easily absorbed [20]. However, also, the low consumption of foods rich in iron; dried beans, dried apricots, prunes, raisins and other dried fruits, almonds, whole grains, and yams [21-26]. Additionally, in this part of the world, the sex of the head of the household is indirectly associated with anaemia in that a family headed by a woman would be more exposed to malnutrition [27,28].
Smoking within the family also seems to be one of these indirect factors [29,30]. In settings where iron deficiency is the most common cause, additional iron is usually provided by iron supplements to vulnerable groups, especially young children from 6 to 24 months and pregnant women [31,32]. Diet-based approaches to increasing iron intake through food fortification and dietary diversification are important and sustainable strategies for preventing iron deficiency anaemia in the general population [33,34]. In settings where iron deficiency is not the sole cause of anaemia, approaches combining iron interventions with other measures are needed [35,36]. Strategies should include controlling other causes of anaemia and should be integrated into the primary health care system and existing programs [37]. These strategies should be adapted to local conditions, considering anaemia's specific etiology and prevalence in a given setting and population group [10,11]. In endemic or epidemic malaria situations, prevention and treatment of the disease will be essential to reduce the prevalence of anaemia [38]. Likewise, when hookworms and other intestinal parasites are endemic, children and women should be given prophylactic deworming drugs [6].
Given the multifactorial nature of this disease, correction of anaemia often requires an integrated approach [38]. Thus, knowing the determinants of anemic children they could allow the health authorities to target these highly vulnerable groups and thus prevent the occurrence of anaemia, or adapt the care strategies for these children. Hence, the general objective of this study was to determine the factors associated with the occurrence of anaemia in children aged from 6 to 59 months in Guinea, and specifically it was also to describe the profile of anaemic children aged 6 to 59 months in Guinea, identify the sociodemographic, clinic and economics factors associated with anaemia among these children.
Study design: a cross-sectional study was conducted to determine risk factors associated with aneamia among children aged 6 to 59 months in the Republic of Guinea.
Study setting and population: the Republic of Guinea is located in the southwest of West Africa (Latitude North 7°30' and 12°30' Longitude West: 8° and 15°) with an area of 245,857 km2. It is bordered by six countries: Guinea-Bissau to the West, Senegal and Mali to the North, Côte d'Ivoire to the East, Liberia and Sierra Leone to the South. It is a coastal country with 333 km of coastline on the Atlantic and extends over 800 km from east to west and 500 km from north to south. Its climate is tropical, alternating between a rainy season and a dry season of about six months each. The country gives rise to the main rivers of the sub-region: the Senegal River, the Niger River, the Gambia River, the Konkouré River, the Kolente River, and the Lofa River. Guinea comprises four natural regions: Lower Guinea, Middle Guinea, Upper Guinea and Forest Guinea. Lower Guinea is a region of coastal plains which covers 18% of the national territory and which is characterized climatically by heavy rainfall varying between 3000 and 4000�mm of water per year with high humidity. Middle Guinea, a region of mountain ranges, covers 22% of the national territory with annual precipitation levels varying between 1500 and 2000 mm with a semi-temperate climate. Upper Guinea is a region of plateaus and wooded savannas covering 40% of the national territory. The last General Census of Population and Housing (RGPH) of 2014 established the population of Guinea at 10,523,361 inhabitants.
According to estimates by the National Institute of Statistics of Guinea, the population increased to 12,559,623 inhabitants in 2020, with an intercensal annual growth rate of 2.9%. Women make up nearly 55% of the population. Most of the population is young (44% are under the age of 15 in 2014). Only 4% of the Guinean population was over 65 in 2014. This population is very young and predominantly rural. The crude birth rate was 33.6 per 1,000 in 2018 and the total fertility rate was 4.8 children in 2018. The average life expectancy at birth is 59 years. The Guinean population presents a great ethnic diversity. The ethnic groups are as follows: the Malinkés, the Koniankés, the Peulhs, the Toucouleurs, the Diakankés, the Soussous, the Bagas, the Nalous, the Mikoforès, the Kissis, the Guerzés, the Tomas, the Manons, the Konos, the Badiarankés, Bassaris, Koniaguis, Landoumas, Lélés, Foulakoundas, Tomamanians, Kourankos, Djallonkés. Islam is the dominant religion in the country (85%). The rest of the population practices Christianity (4.3%) and traditional beliefs. Economically, despite the agricultural, mining and water potential, Guinea remains fragile and vulnerable. Guinea's gross domestic product in 2017 was US$8.8 billion, or US$848 per capita. The inflation rate in 2019 stood at 9.5%. In terms of human development, Guinea was ranked 175th out of 189 countries in 2017 and a poverty rate of 43.7%. The unemployment rate for people over 15 is estimated in 2014 at 5.2%. In terms of education, the country has a gross primary school enrollment rate of 88.6% and a gross secondary school enrollment rate of 29.7% in 2017. The overall literacy rate for those over 15 is estimated at 32% in 20145. Our study population was composed of children aged 6 to 59 months. All children in the selected areas were eligible to be included in the study.
Variables: variables included region, education, head of family gender, literacy, fortified foods, mother´s age, mosquito net, tobacco chew, cigarette, income, twins, residence, maternal aneamia level, ethnic group, feeding with milk, child gender, intestinal worms, vitamin A withing 6 months, iron, maternal age, index of well-being, index of well-being residence zone and level of anaemia. The “Level of anaemia” is the outcomes variable which is qualitative ordinal with four modalities define as: mild anaemia: hemoglobin level between 10.0 and 10.9 grams/deciliter; moderate anaemia: hemoglobin level between 7.0 and 9.9 grams/deciliter; severe anaemia: hemoglobin level below 7.0 grams/deciliter; absence of anaemia: hemoglobin level greater than or equal to 11 ranges/deciliter. We considered possible confounders in our analysis.
Data source: this was a secondary data analysis from Guinea's fifth demographic and health survey (EDS V 2018). These surveys covered the entire national territory and aimed to produce representative results at the level of the whole country, for the city of Conakry, for the other cities, for the urban and rural areas, and at the level of administrative regions of Guinea. Data collection occurred from March to June 2018.
Sample size: we included in this study all children satisfying inclusion criteria (aged between 6 and 59 months).
Data analysis: multiple correspondence factor analysis (MCA) allowed us to describe the aneamic profile of children according to their level of anaemia. In carrying out the MCA, the dependent variable “level of anaemia” was considered illustrative, and the independent variables as explanatory. Given the weighted nature of our data, we used the “svydisign” function of the “survey” package of the R software, to consider our primary units and the associated weighting coefficients, then, we initialized our ordered logistic model with the "svylr" function of the "survey" package of the R software. We used a simple top-down step-by-step procedure for the final selection of the explanatory variables (without the possibility for a variable excluded at a previous step to be included later in the model), starting from the model containing the explanatory variables. potential, while respecting the minimization criterion of the Akaïke (Akaïke Information Criteria (AIC)), thanks to the “stepAIC” function of the “MASS” package. Then, a bottom-up walkthrough to see if the results converge. From the selected model, we looked for source variables of multicollinearity by examining the variance inflation factor (VIF) Using the "vivid" function of the "car" package of the software R. The VIF estimates by how much the variance of a coefficient is increased due to a linear relationship with other explanatory variables. If the VIFs are equal to 1, then there is no multi-collinearity. On the other hand, if some VIFS are greater than 1, the explanatory variables are correlated [38]. However, since there is no consensual threshold from which we can consider that a variable is a source of multicollinearity in a regression model, we considered the threshold of 2.5 as suggested by Paul Allison [39].
For the validation of our model, three statistical tests were used: the parallel regression hypothesis or the proportional odds' hypothesis is necessary for applying the ordinal logistic regression model for an ordered categorical variable. Thus, we tested this hypothesis using a Brant test in R software, which is available in the brant package with the “brant” function. If the P-value was greater than 0.05, this led to the rejection of the null hypothesis and allowed us to conclude that the parallel regression hypothesis was valid or that the coefficients do not differ according to the different cut-off points in the result. Variable; the Wald test to assess the overall significance of our model under the null hypothesis that the beta coefficients of our model were equal and equal to zero; The Hosmer Lemeshow's test for which the number of degrees of freedom was equal to the number of groups minus 2 (number of groups - 2). If P-value <0.05; we rejected the null hypothesis H0. This would mean that our model is not well specified or does not fit well to the data. For the analysis of our data, we used R statistical software version 4.0.3 (2020-10-10). For selecting the variables to be included in the regression model, the risk of type I (risk of error α) was set at 20% and the significance of our final model, α was set at 5%.
Ethical consideration: the approval of an institutional review board was not required for this work. Our data is in the public domain, and we have obtained authorization from The Demographic and Health Surveys whose program is specialized in collecting, analyzing, and disseminating accurate and representative data on population health, through more than 400 surveys in over 90 countries including Guinea.
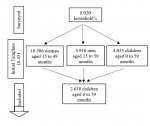
Characteristics of our study population: the female sex was the majority (52.0%). Most of these children resided in urban areas (71.2%) and were of Toma ethnicity (41.5%). More than half of these children did not sleep under mosquito nets (61.6%). Moderate anaemia was found in 41.1% of the children, followed by the severe form (28.6%) and the mild form (28.2%), and only 2.1% had a normal level of anaemia. 82.6% of these children had received a fortified diet, 75.7% iron supplementation, and 56.0% vitamin A intake. The households in which the surveyed children lived are mostly well-off (23.0%) and are headed most often by women (87.6%) and mothers could only read part of the text most often 84.7%. In anemic mothers, the mild form was found in 57.4% of mothers and these mothers did not consume tobacco in any form (99.4%). Figure 1 summarizes the sample selection flow chart.
Treatment of missing data: only 4 variables out of the variables in this study had missing values. In addition, the proportion of these missing data varied between 0.04% and 4.10%. The sensitivity analysis carried out on the single results of 5 complete analyzes, compared to 10 complete analyzes, made it possible to retain 5 imputations.
Characterization of children on the factor axes: we interpreted only the modalities having good contribution to the construction of the axes and the highest squared cosines (well represented on the axes); axis 1 pitted children from the regions of Boké, Faranah, Labé, Mamou, and Nzérékoré with children living in the regions of Conakry and Kindia. It pitted children whose mothers had no education against those whose mothers had attended school. He opposed the Malinké and Toma ethnic groups to the Soussou, Peulh, Kissi, and Guèze ethnic groups. He opposed households headed by a woman to those headed by a man. He pitted children whose mothers could read only part of the text against those whose mothers could read the entire text. He pitted children who had not received fortified foods, whose mothers had no income, lived in poor or well-off households, against those whose mothers were visually impaired or could read the whole text, smoked cigarettes, whose children had taken neither iron nor vitamin A during the last 6 months preceding the survey, lived in rural areas and whose households were wealthy (Table 1). Axis 2 opposed children whose mothers resided in the regions of Faranah, Kankan, Nzérékoré, whose mothers had no education, from Peulh, Malinké, Kissi, and those whose mothers resided in the regions of Boké, Conakry, Kindia and Labé having an education, from Ethnie Toma and Guerzé who could not read a whole text. He pitted children whose mothers were moderately anemic, slept under mosquito nets, paid workers, but who had no memory of fortified food intake, or iron intake, to children who did not sleep under mosquito nets, who did not who had not taken the vitamin A, who lived in rural areas and whose households were well-off (Table 2).
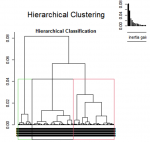
Ascending hierarchical classification (AHC): according to the histogram of the level indices, the best partition is the one made up of 3 classes. Each of the groups has specific characteristics (Figure 2). Class 1 is mainly made up of children whose mothers do not smoke, do not use tobacco or pipes (100%), can read part of the text (95%), are well-off (90%), have no education (90%), of Toma ethnicity (88), whose children received iron supplementation (86%) and a fortified diet (86%), whose head of household is a woman (84%), who do not use a mosquito net (78%), whose children took vitamin A in the last 6 months before the survey (70%) and whose mothers suffered from mild anaemia (60%). Class 2 is mainly made up of children whose mothers do not smoke, nor consume tobacco, nor pipes (100%), can read part of the text (97%), whose head of the family is female (94 %), whose children received a fortified diet (94%), whose mothers had no education (89%) and lived in urban areas (86%), whose children received iron supplementation (71%), mainly of the Kissi ethnic group (58%) and who sleep under a mosquito net (53%). Class 3 is mainly made up of children whose mothers are under 18 (99%), residing in rural areas (88%), children from households where the head of the household is female (82%). Contractual (72%), who were still breastfeeding at the time of the survey (65%) and rich (62%) whose children received fortified food (58%).
Multivariate analysis: thus, the variables significantly explaining anaemia in children from 6 to 59 months in Guinea are: mother´s lack of education [1.81 (1.46-2.33, p = 0.047)], the fact that the mother is blind or visually impaired [4.14 (3.01-7.79, p = 0.007)], or can only read part of the text [3.33 (1.96-4.70, p = 0.017)], the absence of a fortified diet [2.48 (1.98-4.38, p = 0.019)], mother under the age of 18 [6.70 (4.59-7.87, p = 0.035)], does not sleep under the mosquito net [1.78 (1.47-3.58, p = 0.042)], resides in urban area [2.1 (1.57-4.61, p = 0.033], mother anaemia whether severe [2.94 (1.96-3.81, p = 0.047)], moderate [1.25 (1.07-2.74, p = 0.033)] or slight [1.94 (1.96-3.81, p = 0.016)]. Female child [1.73 (1.21-2.11, p = 0.027)], the child has not taken vitamin A during the last 6 months [3.6 (2.50-4.47, p = 0.042)], has no iron supplementation [1.34 (1.01-2.0, p = 0.001)], poorer family [10.30 (1.09-13.6, p = 0.043)], poor [5.63 (4.54-9.69, p = 0.02)] and even modest [2.63 (1.54-3.69, p = 0.04)]. Table 3, Table 3 suite, Table suite 1 and Table 3 suite 2 summarizes determinants of anaemia.
Our study focused on a representative sample of children aged 6 to 59 months in Guinea. In addition to measuring the hemoglobin level, which is the benchmark indicator for diagnosing anaemia in each population; the causes and profiles of anaemia must be investigated, as they may vary from one population to another. Thus, our study made it possible to identify three (03) clusters of anemic children with common characteristics but also specific to each cluster (the mother's age, the level of education of the mother and the use of mosquito nets. Eleven (11) social, demographic, and economic factors have been identified explaining the occurrence of anaemia in this particularly at-risk age group. Children from underaged mothers are seven (07) times more likely to be anemic compared to those from mothers over. Several hypotheses can explain this result: First, early pregnancy or early marriage in the context of poverty and rural areas. Secondly, pregnancies outside marriage expose these women to less attention in a context marked by religious. Likewise, children born to poor mothers are six (06) times more likely to be anemic compared to those who are rich, this can be explained by the fact that poverty is an indirect factor of anaemia because it causes malnutrition, leading to the onset of anaemia [40]. Children whose mothers are blind or partially sighted are four times more likely to be anemic than those who are not In Guinea, most awareness-raising campaigns are carried out via textual media such as banners, mobile notifications and even text messages looping over national television stations. This immediately deprives this group of mothers of essential information for adequate nutrition for their children.
On the other hand, children residing in urban areas are twice (02) times more likely to be anemic compared to children residing in rural areas, This was until now unrecognized by the health authorities and could be explained by the diet rich and accessible in vegetables and legumes for children living in rural areas compared to those in urban areas where the high demographic concentration exposes them to the high cost of living and a diet low in nutrients rich in iron, which directly impacts the nutrition of the populations living there and indirectly on the anemic status of children. Children who did not receive vitamin A supplementation were four (04) more likely to be anemic compared to children who received supplementation [3.6 (2.50-4.47, p = 0.042)]. This can be explained by the WHO´s policy regarding vitamin A supplementation in children, starting at the age of 6 months, as a public health intervention to reduce infant mortality and morbidity; it is a periodic activity which sometimes suffers from shortcomings, especially in terms of coverage, as highlighted by the non-governmental organization Helen Keller International in their 2018 “Annual report on vitamin A supplementation in Guinea”. Lack of education among mothers exposes two (02) times more children to the occurrence of anaemia [1.81 (1.46-2.33, p = 0.047)]. Indeed, despite initiatives such as Girl´s Education Acceleration Project (GEAP), Promotion of girl´s participation in school activities through the establishment, training, and support for Operation Government of Children (GDE) in schools launched by UNICEF in 2015 in the prefecture of Telimélé, the enrollment of young women remains very low in Guinea with the main consequence of the inability to access essential information essential in terms of nutrition.
While our study has strengths, it also has some weaknesses. Among the strengths are: the source of our data from the fifth Demographic and Health Survey in Guinea (EDS V) carried out every 5 years on average by the National Institute of Statistics (INS), the Ministry of Planning and Economic Development of Guinea, in collaboration with the Ministry of Health with the financial support of the Guinean Government. Second, the representativeness of our sample. Indeed, the 2018 DHS sample is a stratified random sample drawn at two stages that covered the entire national territory. Finally, using a multiple imputation approach to treat missing data is an unbiased method in contrast to the simple imputation approach [41]. However, our study was carried out retrospectively, which constitutes a limitation insofar as certain information demonstrated in the literature as associated with the occurrence of anaemia in other populations were not considered in our model of logistic regression. Then our model explains 68.3% of the anaemia, which, although satisfactory, still constitutes a loss of 31.7% of the information, which, although low, constitutes a significant part of information that could be recovered in study prospects.
Through this work, we were able to show that in Guinea, the anaemic profile of children from 6 to 59 months was different according to the mother's age, the level of education of the mother and the use of the impregnated mosquito net. Likewise, although in Guinea, most of the explanatory factors of anaemia are common to those found in certain countries of sub-Saharan Africa, the mechanisms of action of these explanatory factors are quite different between children suffering only from anaemia and mild, moderate, or severe anaemia. Therefore, these differences can be used to establish anaemia. Therefore, these differences can be used to establish well-targeted public health policies for the control of anaemia in all its forms.
What is known about this topic
- Mother´s lack of education, the mother low literacy level, the absence of a fortified diet, mother under the age of 18, does not sleep under the mosquito net, mother anaemia whether severe, moderate or slight. Female child, the child has not taken vitamin A during the last 6 months, has no iron supplementation, poorer family, poor and even modest are associated with aneamia.
What this study adds
- We found in our study that leaving in an urban area was also associate with anaemia occurrence among children in Guinea;
- Tree (03) clusters of anaemic children have been identified: The first one with the specific features of the absence of the use of an impregnated mosquito net and the presence of mild anaemia in the mother. The second one is made up of mothers living mainly in urban areas, and the third one is made up of mothers under the age of 18.
The authors declare no competing interests.
All authors contributed to the study conception and design. Kuotu had the idea for the article and performed the literature search and statistical analysis, Toure and Magassouba drafted the article, and Doumbouyah and Toure critically revised the work. All the authors have read and agreed to the final manuscript.
Table 1: modalities that contribute the most to the construction of axis 1
Table 2: modalities that contribute the most to the construction of axis 2
Table 3: explanatory factors for anaemia in children aged 6 to 59 months in Guinea
Table 3 suite: explanatory factors for anaemia in children aged 6 to 59 months in Guinea
Table 3 suite 1: explanatory factors for anaemia in children aged 6 to 59 months in Guinea
Table 3 suite 2: explanatory factors for anaemia in children aged 6 to 59 months in Guinea
Figure 1: sample selection flow chart
Figure 2: ascending hierarchical classification (AHC)
- DNS/Guinée DN de la S-, Macro ORC. Guinée Enquête Démographique et de Santé 2005. 2006. Accessed Oct 22, 2020.
- UNICEF Guinée. Enquête Démographique et de Santé 2018. Accessed Aug 26, 2020.
- WHO. The world health report 2000 - Health systems: improving performance. Accessed Jan 30, 2020.
- Baker WF, Lee R. Iron deficiency, folate, and vitamin B12 deficiency in pregnancy, obstetrics, and gynecology. In: Frenkel EP, Sarode R, Bick RL, Baker WF, éditeurs. Hematological Complications in Obstetrics, Pregnancy, and Gynecology. Cambridge: Cambridge University Press; 2006. Accessed August 26, 2020.
- Sumbele IUN, Samje M, Nkuo-Akenji T. A longitudinal study on anaemia in children with Plasmodium falciparum infection in the Mount Cameroon region: prevalence, risk factors and perceptions by caregivers. BMC Infect Dis. 5 mars 2013;13:123. PubMed | Google Scholar
- Lynch S, Pfeiffer CM, Georgieff MK, Brittenham G, Fairweather-Tait S, Hurrell RF et al. Biomarkers of Nutrition for Development (BOND)-Iron Review. J Nutr. 01 2018;148(suppl_1):1001S-1067S. PubMed | Google Scholar
- Wessells KR, Hess SY, Ouédraogo ZP, Rouamba N, Ouédraogo J-B, Brown KH. Asymptomatic malaria infection affects the interpretation of biomarkers of iron and vitamin A status, even after adjusting for systemic inflammation, but does not affect plasma zinc concentrations among young children in Burkina Faso. J Nutr. 2014 Dec;144(12):2050-8. PubMed | Google Scholar
- Khan S, Zaheer S, Safdar NF. Determinants of stunting, underweight and wasting among children < 5 years of age: evidence from 2012-2013 Pakistan demographic and health survey. BMC Public Health. 2019 Apr 1;19(1):358. PubMed | Google Scholar
- Awasthi S, Bundy DAP, Savioli L. Helminthic infections. BMJ. 2003 Aug 23;327(7412):431-3. PubMed | Google Scholar
- Sumbele IUN, Nkemnji GB, Kimbi HK. Soil-transmitted helminths and plasmodium falciparum malaria among individuals living in different agroecosystems in two rural communities in the mount Cameroon area: a cross-sectional study. Infect Dis Poverty. 2017 Mar 16;6(1):67. PubMed | Google Scholar
- Black RE, Victora CG, Walker SP, Bhutta ZA, Christian P, de Onis M et al. Maternal and child undernutrition and overweight in low-income and middle-income countries. Lancet. 2013 Aug 3;382(9890):427-45. PubMed | Google Scholar
- Institute of Medicine (US) Committee on Nutritional Status During Pregnancy and Lactation. Washington (DC). Nutrition During Pregnancy: Part I Weight Gain: Part II Nutrient Supplements. National Academies Press (US). 1990. Accessed Aug 26, 2020.
- Calis JCJ, Phiri KS, Faragher EB, Brabin BJ, Bates I, Cuevas LE et al. Severe anaemia in Malawian children. N Engl J Med. 2008 Feb 28;358(9):888-99. PubMed | Google Scholar
- Mabli J, Ohls J. Supplemental Nutrition Assistance Program participation is associated with an increase in household food security in a national evaluation. J Nutr. 2015 Feb;145(2):344-51. PubMed | Google Scholar
- Mabli J, Worthington J. Supplemental nutrition assistance program participation and child food security. Pediatrics. 2014 Apr;133(4):610-9. PubMed | Google Scholar
- Rivera RL, Maulding MK, Abbott AR, Craig BA, Eicher-Miller HA. SNAP-Ed (Supplemental Nutrition Assistance Program-Education) Increases Long-Term Food Security among Indiana Households with Children in a Randomized Controlled Study. J Nutr. 2016 Nov;146(11):2375-2382. PubMed | Google Scholar
- Bliznashka L, Arsenault JE, Becquey E, Ruel MT, Olney DK. Using structural equation modelling to understand the contributors to anaemia among young Burkinabe children. Matern Child Nutr. 2020;16(1):e12881. PubMed | Google Scholar
- Tarasuk V, Fafard St-Germain A-A, Mitchell A. Geographic and socio-demographic predictors of household food insecurity in Canada, 2011-12. BMC Public Health. 2019 Jan 3;19(1):12. PubMed | Google Scholar
- Kursmark M, Weitzman M. Recent findings concerning childhood food insecurity. Curr Opin Clin Nutr Metab Care. 2009 May;12(3):310-6. PubMed | Google Scholar
- Belachew T, Lindstrom D, Gebremariam A, Jira C, Hattori MK, Lachat C et al. Predictors of chronic food insecurity among adolescents in Southwest Ethiopia: a longitudinal study. BMC Public Health. 2012;12:604. PubMed | Google Scholar
- Christian AK, Wilson ML, Aryeetey RNO, Jones AD. Livestock ownership, household food security and childhood anaemia in rural Ghana. PloS One. 2019;14(7):e0219310. PubMed | Google Scholar
- Slater HC, Foy BD, Kobylinski K, Chaccour C, Watson OJ, Hellewell J et al. Ivermectin as a novel complementary malaria control tool to reduce incidence and prevalence: a modelling study. Lancet Infect Dis. 2020 Apr;20(4):498-508 PubMed | Google Scholar
- McDonald CM, McLean J, Kroeun H, Talukder A, Lynd LD, Green TJ. Household food insecurity and dietary diversity as correlates of maternal and child undernutrition in rural Cambodia. Eur J Clin Nutr. 2015 Feb;69(2):242-6. PubMed | Google Scholar
- Yang Q, Yuan T, Yang L, Zou J, Ji M, Zhang Y et al. Household Food Insecurity, Dietary Diversity, Stunting, and Anaemia among Left-Behind Children in Poor Rural Areas of China. Int J Environ Res Public Health. 2019 Nov 28;16(23):4778. PubMed | Google Scholar
- Marin-Leon L, Francisco PMSB, Segall-Corrêa AM, Panigassi G. [Household appliances and food insecurity: gender, referred skin color and socioeconomic differences]. Rev Bras Epidemiol. 2011 Sep;14(3):398-410. PubMed | Google Scholar
- Negesse A, Jara D, Habtamu Temesgen null, Dessie G, Getaneh T, Mulugeta H et al. The impact of being of the female gender for household head on the prevalence of food insecurity in Ethiopia: a systematic-review and meta-analysis. Public Health Rev. 2020 Jun 5;41:15. PubMed | Google Scholar
- Jung NM, de Bairros FS, Pattussi MP, Pauli S, Neutzling MB. Gender differences in the prevalence of household food insecurity: a systematic review and meta-analysis. Public Health Nutr. 2017 Apr;20(5):902-916. PubMed | Google Scholar
- Sreeramareddy CT, Ramakrishnareddy N, Subramaniam M. Association between household food access insecurity and nutritional status indicators among children aged <5 years in Nepal: results from a national, cross-sectional household survey. Public Health Nutr. 2015 Nov;18(16):2906-14. PubMed | Google Scholar
- Semba RD, Campbell AA, Sun K, de Pee S, Akhter N, Moench-Pfanner R et al. Paternal smoking is associated with greater food insecurity among poor families in rural Indonesia. Asia Pac J Clin Nutr. 2011;20(4):618-23. PubMed | Google Scholar
- Best CM, Sun K, de Pee S, Sari M, Bloem MW, Semba RD. Paternal smoking and increased risk of child malnutrition among families in rural Indonesia. Tob Control. 2008 Feb;17(1):38-45. PubMed | Google Scholar
- Hashizume M, Kunii O, Sasaki S, Shimoda T, Wakai S, Mazhitova Z et al. anaemia and iron deficiency among schoolchildren in the Aral Sea region, Kazakhstan. J Trop Pediatr. 2003 Jun;49(3):172-7. PubMed | Google Scholar
- Akhwale WS, Lum JK, Kaneko A, Eto H, Obonyo C, Bj�rkman A et al. anaemia and malaria at different altitudes in the western highlands of Kenya. Acta Trop. 2004 Jul;91(2):167-75. PubMed | Google Scholar
- Nair KM, Fernandez-Rao S, Nagalla B, Kankipati RV, Punjal R, Augustine LF et al. Characterisation of anaemia and associated factors among infants and pre-schoolers from rural India. Public Health Nutr. 2016 Apr;19(5):861-71. PubMed | Google Scholar
- Ndemwa M, Wanyua S, Kaneko S, Karama M, Anselimo M. Nutritional status and association of demographic characteristics with malnutrition among children less than 24 months in Kwale County, Kenya. Pan Afr Med J. 2017 Nov 24;28:265. PubMed | Google Scholar
- Ahmed F, Prendiville N, Narayan A. Micronutrient deficiencies among children and women in Bangladesh: progress and challenges. J Nutr Sci. 2017 Jan 3;5:e46. PubMed | Google Scholar
- Fiorentino M, Perignon M, Kuong K, Groot R de, Parker M, Burja K et al. Effect of multi-micronutrient-fortified rice on cognitive performance depends on premix composition and cognitive function tested: results of an effectiveness study in Cambodian schoolchildren. Public Health Nutr. 2018 Mar;21(4):816-827. PubMed | Google Scholar
- Huo J, Sun J, Fang Z, Chang S, Zhao L, Fu P et al. Effect of Home-Based Complementary Food Fortification on Prevalence of anaemia Among Infants and Young Children Aged 6 to 23 Months in Poor Rural Regions of China. Food Nutr Bull. 2015 Dec;36(4):405-14. PubMed | Google Scholar
- De-Regil LM, Suchdev PS, Vist GE, Walleser S, Peña-Rosas JP. Home fortification of foods with multiple micronutrient powders for health and nutrition in children under two years of age (Review). Evid-Based Child Health Cochrane Rev J. 2013 Jan;8(1):112-201. PubMed | Google Scholar
- Suchdev PS, Peña-Rosas JP, De-Regil LM. Multiple micronutrient powders for home (point-of-use) fortification of foods in pregnant women. Cochrane Database Syst Rev. 2015 Jun 19;(6):CD011158. PubMed | Google Scholar
- Korenromp EL, Armstrong-Schellenberg JRM, Williams BG, Nahlen BL, Snow RW. Impact of malaria control on childhood anaemia in Africa -- a quantitative review. Trop Med Int Health. 2004 Oct;9(10):1050-65. PubMed | Google Scholar
- Surbek D, Vial Y, Girard T, Breymann C, Bencaiova GA, Baud D et al. Patient blood management (PBM) in pregnancy and childbirth: literature review and expert opinion. Arch Gynecol Obstet. 2020 Feb;301(2):627-641. PubMed | Google Scholar